铝三角铁三角化学方程式总结修改版
- 格式:doc
- 大小:160.00 KB
- 文档页数:6


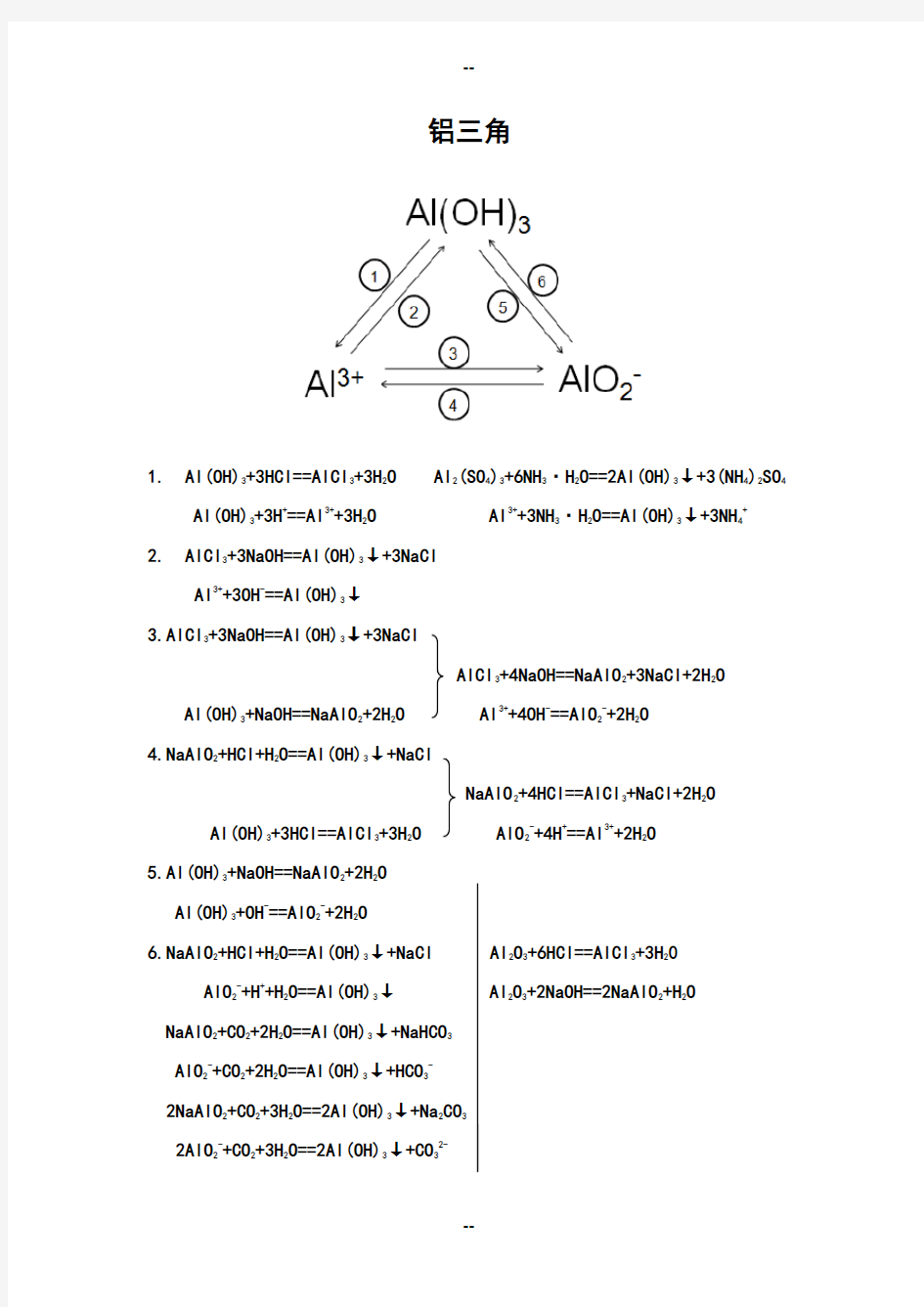
铝三角
1. Al(OH)
3+3HCl==AlCl
3
+3H
2
O Al
2
(SO
4
)
3
+6NH
3
·H
2
O==2Al(OH)
3
↓+3(NH
4
)
2
SO
4
Al(OH)
3+3H+==Al3++3H
2
O Al3++3NH
3
·H
2
O==Al(OH)
3
↓+3NH
4
+
2. AlCl
3+3NaOH==Al(OH)
3
↓+3NaCl
Al3++3OH-==Al(OH)
3
↓
3.AlCl
3+3NaOH==Al(OH)
3
↓+3NaCl
AlCl
3+4NaOH==NaAlO
2
+3NaCl+2H
2
O
Al(OH)
3+NaOH==NaAlO
2
+2H
2
O Al3++4OH-==AlO
2
-+2H
2
O
4.NaAlO
2+HCl+H
2
O==Al(OH)
3
↓+NaCl
NaAlO
2+4HCl==AlCl
3
+NaCl+2H
2
O
Al(OH)
3+3HCl==AlCl
3
+3H
2
O AlO
2
-+4H+==Al3++2H
2
O
5.Al(OH)
3+NaOH==NaAlO
2
+2H
2
O
Al(OH)
3+OH-==AlO
2
-+2H
2
O
6.NaAlO
2+HCl+H
2
O==Al(OH)
3
↓+NaCl Al
2
O
3
+6HCl==AlCl
3
+3H
2
AlO
2-+H++H
2
O==Al(OH)
3
↓Al
2
O
3
+2NaOH==2NaAlO
2
+H
2
O
NaAlO
2+CO
2
+2H
2
O==Al(OH)
3
↓+NaHCO
3
AlO
2-+CO
2
+2H
2
O==Al(OH)
3
↓+HCO
3
-
2NaAlO
2+CO
2
+3H
2
O==2Al(OH)
3
↓+Na
2
CO
3
2AlO
2-+CO
2
+3H
2
O==2Al(OH)
3
↓+CO
3
2-
铁三角
1.Fe+2HCl==FeCl
2+H
2
↑2Na+2H
2
O==2NaOH+H
2
↑
Fe+2H+==Fe2++H
2↑3Fe+4H
2
O(g ) 高温Fe
3
O
4
+4H
2
2.FeCl
2+Zn==ZnCl
2
+Fe Na投入到FeCl
3
溶液中
Fe2++Zn==Zn2++Fe 2Na+2H
2O==2NaOH+H
2
↑
3.2FeCl
2+Cl
2
==2FeCl
3
3NaOH+FeCl
3
==Fe(OH)
3
↓+3NaCl
2Fe2++Cl
2==2Fe3++2Cl- 6Na+6H
2
O+2FeCl
3
==2Fe(OH)
3
↓+6NaCl+3H
2
↑
4.2FeCl
3+Fe==3FeCl
2
2Na
2
O
2
+2H
2
O == 4NaOH+O
2
↑
2Fe3++Fe==3Fe2+ 2Na
2O
2
+2CO
2
==2Na
2
CO
3
+O
2
5.2Fe+3Cl
2 点燃2FeCl
3
2Na
2
O
2
+4HCl==4NaCl+2H
2
O+O
2
↑
6.Fe
2O
3
+3CO ==2Fe+3CO
2
2Al+2NaOH+2H
2
O==2NaAlO
2
+3H
2
↑
Na
2O+H
2
O==2NaOH
Na
2O+CO
2
==Na
2
CO
3
FeCl
3+3NaOH==Fe(OH)
3
↓+3NaCl Na
2
O+2HCl==2NaCl+H
2
O
FeSO
4+2NaOH==Fe(OH)
2
↓+Na
2
SO
4
2NaHCO
3
== Na
2
CO
3
+H
2
O+CO
2
↑
4Fe(OH)
2+O
2
+2H
2
O==4Fe(OH)
3
NaHCO
3
+HCl==NaCl+H
2
O+CO
2
↑
2Fe(OH)
3 ==Fe
2
O
3
+3H
2
O Na
2
CO
3
+2HCl==2NaCl+H
2
O+CO
2
↑
Fe
2O
3
+6HCl==2FeCl
3
+3H
2
0 Na
2
CO
3
+H
2
O+CO
2
==2NaHCO
3
Fe
3O
4
+8HCl==2FeCl
3
+FeCl
2
+4H
2
O NaHCO
3
+NaOH==Na
2
CO
3
+H
2
O
Na
2CO
3
+CaCl
2
==CaCO
3
↓+2NaCl
Na
2CO
3
+BaCl
2
==BaCO
3
↓+2NaCl