《材料科学基础》课后答案(1-7章)
- 格式:doc
- 大小:791.50 KB
- 文档页数:10

武汉理工大学材料科学基础(第2版)课后习题和答案第一章绪论1、仔细观察一下白炽灯泡,会发现有多少种不同的材料?每种材料需要何种热学、电学性质?2、为什么金属具有良好的导电性和导热性?3、为什么陶瓷、聚合物通常是绝缘体?4、铝原子的质量是多少?若铝的密度为2.7g/cm3,计算1mm3中有多少原子?5、为了防止碰撞造成纽折,汽车的挡板可有装甲制造,但实际应用中为何不如此设计?说出至少三种理由。
6、描述不同材料常用的加工方法。
7、叙述金属材料的类型及其分类依据。
8、试将下列材料按金属、陶瓷、聚合物或复合材料进行分类:黄铜钢筋混凝土橡胶氯化钠铅-锡焊料沥青环氧树脂镁合金碳化硅混凝土石墨玻璃钢9、Al2O3陶瓷既牢固又坚硬且耐磨,为什么不用Al2O3制造铁锤?第二章晶体结构1、解释下列概念晶系、晶胞、晶胞参数、空间点阵、米勒指数(晶面指数)、离子晶体的晶格能、原子半径与离子半径、配位数、离子极化、同质多晶与类质同晶、正尖晶石与反正尖晶石、反萤石结构、铁电效应、压电效应.2、(1)一晶面在x、y、z轴上的截距分别为2a、3b、6c,求出该晶面的米勒指数;(2)一晶面在x、y、z 轴上的截距分别为a/3、b/2、c,求出该晶面的米勒指数。
3、在立方晶系的晶胞中画出下列米勒指数的晶面和晶向:(001)与[210],(111)与[112],(110)与[111],(322)与[236],(257)与[111],(123)与[121],(102),(112),(213),[110],[111],[120],[321]4、写出面心立方格子的单位平行六面体上所有结点的坐标。
5、已知Mg2+半径为0.072nm,O2-半径为0.140nm,计算MgO晶体结构的堆积系数与密度。
6、计算体心立方、面心立方、密排六方晶胞中的原子数、配位数、堆积系数。
7、从理论计算公式计算NaC1与MgO的晶格能。
MgO的熔点为2800℃,NaC1为80l℃, 请说明这种差别的原因。

《材料科学基础》课后习题答案第一章材料结构的基本知识4. 简述一次键和二次键区别答:根据结合力的强弱可把结合键分成一次键和二次键两大类。
其中一次键的结合力较强,包括离子键、共价键和金属键。
一次键的三种结合方式都是依靠外壳层电子转移或共享以形成稳定的电子壳层,从而使原子间相互结合起来。
二次键的结合力较弱,包括范德瓦耳斯键和氢键。
二次键是一种在原子和分子之间,由诱导或永久电偶相互作用而产生的一种副键。
6. 为什么金属键结合的固体材料的密度比离子键或共价键固体为高?答:材料的密度与结合键类型有关。
一般金属键结合的固体材料的高密度有两个原因:(1)金属元素有较高的相对原子质量;(2)金属键的结合方式没有方向性,因此金属原子总是趋于密集排列。
相反,对于离子键或共价键结合的材料,原子排列不可能很致密。
共价键结合时,相邻原子的个数要受到共价键数目的限制;离子键结合时,则要满足正、负离子间电荷平衡的要求,它们的相邻原子数都不如金属多,因此离子键或共价键结合的材料密度较低。
9. 什么是单相组织?什么是两相组织?以它们为例说明显微组织的含义以及显微组织对性能的影响。
答:单相组织,顾名思义是具有单一相的组织。
即所有晶粒的化学组成相同,晶体结构也相同。
两相组织是指具有两相的组织。
单相组织特征的主要有晶粒尺寸及形状。
晶粒尺寸对材料性能有重要的影响,细化晶粒可以明显地提高材料的强度,改善材料的塑性和韧性。
单相组织中,根据各方向生长条件的不同,会生成等轴晶和柱状晶。
等轴晶的材料各方向上性能接近,而柱状晶则在各个方向上表现出性能的差异。
对于两相组织,如果两个相的晶粒尺度相当,两者均匀地交替分布,此时合金的力学性能取决于两个相或者两种相或两种组织组成物的相对量及各自的性能。
如果两个相的晶粒尺度相差甚远,其中尺寸较细的相以球状、点状、片状或针状等形态弥散地分布于另一相晶粒的基体内。
如果弥散相的硬度明显高于基体相,则将显著提高材料的强度,同时降低材料的塑韧性。

SOLUTION FOR CHAPTER 71. FIND: Label the phase fields in Figure HP7-1.SOLUTION: There are two regions of single phase equilibrium separated by a region of two-phase equilibrium. The line separating the single phase liquid from the two phaseliquid and solid region is called the liquidus, and the line separating the two phase liquid and solid from the single phase solid is called the solidus.SKETCH:2. FIND: The name given to the type of equilibrium diagram shown in Fig. HP7-1.SOLUTION: The diagram is termed isomorphous. Above the liquidus there is only one phase, namely the liquid, and it has the same structure regardless of composition. Below the solidus is the solid phase, and as well it has the same structure regardless ofcomposition.3. FIND: What is the crystal structure of component B, why?SOLUTION: There are no phase boundaries below the solidus consequently the phase and therefore the structure must be the same. If A is FCC, then B and all compositionsbetween A and B must be the same phase and hence same structure. If A is FCC, then B must also be FCC.4. FIND: Sketch equilibrium cooling curves for alloy X o and pure component B. Explainwhy they have different shapes.SOLUTION: The slope of the temperature versus time behavior for the alloy in thesingle phase region is controlled by the cooling rate and the heat transferred from theliquid. At the liquidus temperature a small amount of solid is formed, releasing anamount of latent heat of fusion related to the volume of solid transformed. Hence, at the liquidus temperature there will be a change in slope. Since heat is being generated theslope will be less than that above the liquidus. As the temperature is reduced through the two-phase region a small amount of solid is formed and a corresponding heat released;once the solidus temperature is reached no additional transformation takes place and there is again a change in slope. The slope is greater as the solid cools than as the liquid andsolid cooled. For the pure component, solid and liquid are in equilibrium at T B, and hence,a horizontal line when liquid transforms to solid at T B.SKETCH:5. FIND: The liquidus temperature and the solidus temperature of alloy X oSOLUTION: From figure HP7-1 the liquidus temperature is approximately 1110o C and the solidus temperature is approximately 1070o C.6. FIND: Determine compositions and phase fractions of each phase in equilibrium at1100o C for alloy X o.SOLUTION: At equilibrium, the temperature of two phases must be the same, and thecomposition of the solid is found where the tie line intersects the solidus and the liquid is at the intersection of the tie line with the liquidus. From the phase diagram thecomposition of solid is 0.35B and that of the liquid is 0.55B. Using the lever rule thefraction of liquid, f L, and fraction of solid, f s is determined.7. FIND: Sketch f L and f S for alloy X o as the alloy is cooled under equilibrium conditionsfrom 1200o C to room temperature.SOLUTION: At the liquidus temperature the amount of liquid is almost 100% with onlya very small amount of solid. Conversely at the solidus temperature, the amount of solidis almost 100% with only a very small amount of liquid. In a two-phase field:f L + f s = 1.8. FIND: Changes in the compositions of the liquid and solid phases during quilibriumcooling of alloy X o through the two-phase field.SOLUTION: At the liquidus temperature, the composition of the solid is approximately0.30B. As equilibrium solidification progresses, the composition of the solid increases to0.5 B, the maximum value it can reach. The liquid on the other hand is initially thecomposition of the alloy X o, and as equilibrium solidification progresses the composition increases in B to the maximum composition in the liquid of approximately 0.7 B.9. FIND:From the following data construct a plausible equilibrium phase diagram.Component A melts at 800︒C and B melts at 1000︒C; A and B are completely soluble in one another at room temperature; and if solid α containing 0.3 B is heated underequilibrium conditions, the solid transforms to liquid having the same composition at500︒C.GIVEN: Melting temperatures for the two components, congruent melting temperature and composition.SOLUTION: The sketch shown below satisfies all of the requirements stated in theproblem. The melting temperature of pure A is 800︒C and that for pure B is 1000︒C. The congruent melting temperature is at 500︒C at a composition containing 0.3 B. Below the congruent melting temperature there is a continuous α solid solution.10. FIND: For alloys containing 10, 22, 25, 27, and 40 wt% Ni, determine the number ofphases present and the composition of the phases in equilibrium at 1200o C.11. FIND: Beginning with a statement of mass balance, derive the lever rule in a two-phasesystem.SOLUTION: In a two phase field for an alloy of some over all composition X o, thesolute is distributed in the two phases: x o = fα xα + fβ xβ where the compositions areexpressed in terms of one of the components, fα + fβ = 1. Then, fα= 1 - fβ. Substituting:x o = (1 - fβ) xα + fβ xβx o = xα - xα fβ + xβfβx o - xα = (xβ - xα) fβ12. FIND: Discuss each of the factors that permit the Cu-Ni system to be isomorphous overthe temperature range 350-1000o C.SOLUTION: The empirical rules of Hume-Rothery identify the characteristics that two elements must have in common for extensive solubility. This should require thata. the two components must have the same crystal structureb. the atomic radii of the two atoms must be similarc. the two components have the comparable electro negativities, andd. the two components have the similar valence.13. FIND: What does the temperature 322O C represent in Figure HP7-2?SOLUTION: 322o C is called the critical temperature. At the critical temperature there is a corresponding critical composition. For an alloy of this composition, cooling under equilibrium conditions from above the critical temperature to below this temperatureresults in the formation of two phases from one phase of the critical composition. Ascooling continues the two phases that form have different compositions.14. FIND: In Figure HP7-2, are α1 and α2 different crystal structures?SOLUTION: α1 and α2 are two phases having the same structure but differentcompositions. The composition of the two phases are determined as in any two phasesystem by using the tie-line. Where the tie-line at the equilibrium temperature intersects the phase boundaries determines the composition of the two phase in equilibrium.15. FIND: Location of the equilibrium phase boundary at temperature T.GIVEN: Alloy 1 containing 30%B and alloy 2 containing 50% B, when equilibrated at temperature T are in the same two-phase (L + S) region. The fraction of liquid in alloy 1 is 0.8 and the fraction of liquid in alloy 2 is 0.4.SKETCH:SOLUTION: Since the fraction of liquid in alloy 1 is greater than that of solid, theliquidus is to the left of 0.3, and since the fraction of solid in alloy 2 is greater than that of liquid, the solidus must lie to the right of alloy 2. Using the lever rule:for alloy 1for alloy 20.8X L - 0.8X s = X s - 0.30.4X L - 0.4X s = X s - 0.5Solve for X s and X L,0.8X s - 0.8X L = X s - 0.30.8X s - 0.8X L = 2X s - 1.0____________________0 = -X s + 0.7, orX s = 0.7BTo find X L, substitute X s into one of the equations:0.4 - 0.56 = -0.8X L-0.16 = -0.8X L, orX L = 0.2BAs a check, use the other equation to calculate the fraction of liquid from thecompositions.16. FIND: Label all regions of the phase diagram and the boundaries of monovariantequilibrium for the diagram shown in Figure HP7-3.SKETCH/SOLUTION:17. FIND: Sketch an equilibrium cooling curve from above the eutectic to roomtemperature for an alloy of eutectic composition.SKETCH/SOLUTION:18. FIND: Explain why the equilibrium cooling curves for alloys on either side of theeutectic composition will be different than the equilibrium cooling curve for a eutecticalloy.SOLUTION: At the eutectic temperature, liquid of composition X E is in equilibriumwith two solids, Xα and Xβ . For a hypoeutectic alloy (composition to the left of theeutectic), the first phase to form at the liquidus temperature of the alloy is α. When the eutectic temperature is reached the liquid of the eutectic composition is in equilibriumwith the two solids, one of composition Xα and the other Xβ. Consequently, depending upon composition, the closer the overall composition of the alloy is to X E, the lessproeutectic α and the more eutectic. Similarly, for alloys to the right, proeutectic β will form. In terms of the phase rule at constant pressure, for the eutectic reactionF = C - P + 1 = 2 - 3 + 1 = 0. The eutectic is invariant, and solidifies under equilibriumconditions at one temperature, T E. For alloys on either side of the eutectic composition but between Xα and Xβ, the proeutectic phase forms and cooling occurs as it does in any two-phase s-l region. Once the eutectic isotherm is reached, the remaining liquid ofeutectic composition solidifies isothermally.19. FIND: The maximum solid solubility of B in A and of A in B in Figure HP7-3.SOLUTION: The maximum solubility of B in A occurs at 0.15 B and the maximumsolubility of A in B is 0.1 A.20. FIND: For an alloy of eutectic composition in Figure HP7-3, determine the compositionof the solid phases in equilibrium with the liquid.SOLUTION: The composition of the two solid phase α and β in equilibrium with liquid at T_ occurs at 0.15 B and 0.9 B.21. FIND: Plot f L, fα, and fβ as a function of temperature for the equilibrium cooling of analloy of eutectic composition.SOLUTION: At just above the eutectic temperature, the microstructure consists of allliquid of composition X E. (Determined from the phase diagram to be approximately0.63B.) Just below the eutectic temperature, the microstructure is a mixture of twophases, α and β. From the phase diagram, Xα ~ 0.15B and Xβ ~ 0.9B. To determine the fractions of α and β at temperatures below T E, use the lever-rule. Tabulated below arecompositions of the phases in equilibrium at several temperatures estimated from thephase diagram. These compositions are then used to calculate the amount of each phase present.Fraction of phases:At 600: f L = 1.0, fα = fβ = 0At 500: f L = 0Instal l Equa tion E ditor a nd do uble-click h ere to view equat ion.Instal l Equa tion E ditor a nd do uble-click h ere to view equat ion.At 400: f L = 0Instal l Equa tion E ditor a nd do uble-click h ere to view equat ion.Instal l Equa tion E ditor a nd do uble-click h ere to view equat ion.At 300: f L = 0Instal l Equa tion E ditor a nd do uble-click h ere to view equat ion.Instal l Equa tion E ditor a nd do uble-click h ere to view equat ion.22. FIND: For alloy X o (in Figure HP7-3), calculate the fraction of α that forms as primary αand the fraction of α that forms by eutectic decomposition when the alloy is cooled from 850o C to room temperature under equilibrium conditions.SOLUTIONS: The fraction of proeutectic α that forms under equilibrium conditionspresent in alloy X o cooled from 850o C to just above the eutectic is:Then the fraction of liquid which is of eutectic composition is:f L = 1 - fα = 1. - 0.69 = 0.31.The fraction of α that forms from liquid of eutectic composition, fα,E is :fα,E = (fraction of α formed from eutectic) (0.31)To check these results, the sum of proeutectic α plus that which forms from the eutectic =0.69 + 0.11 = 0.8. That amount should be the same as if we calculated directly fα that would be present just below the eutectic for alloy of composition X o.23. FIND: The total fraction of α at room temperature for alloy X o in Figure HP7-3.SOLUTION: Solubility of B in α at room temperature is approximately 0.02 A and the solubility of A in β is approximately 0.04 A (in terms of B 0.96 B).24. FIND: Equilibrium phase diagram given the information below.GIVEN: Component A melts at 900o C; component B melts at 1000o C; and there is aninvariant reaction at 600o C. The solubility of B in α is known to increase from almost nil at room temperature to a maximum of 10%. When an alloy containing 30% B is cooled under equilibrium conditions just above 600o C, a two-phase mixture is present, 50% αand 50% liquid. When the alloy is cooled just below 600o C, the alloy contains two solid phases, α and β. The fraction of α is 0.75. After cooling under equilibrium conditions to room temperature, the amount of α in the α + β mixture decreases to 68%.SOLUTION: At just above the invariant temperature there is a region of two phaseequilibrium, a mixture of 50% α and 50% L. Consequently if the overall composition is 50%β, and α is located at 10%β, then the liquid must be at ˜50% B. Just below theinvariant reaction, two solid phases are present, αat ˜10%B and β at some composition, Xβ. If fα = 0.75, then:At room temperature, if fα = 0.68, and the solubility of B in α is nil, thenThe invariant reaction is L _ α + β, a eutectic, hence the diagram shown below fits allconditions.25. FIND: A binary phase diagram consistent with the information given below.GIVEN: The binary system A-B with T B > T A is known to contain two invariantreactions of the type: L _ α + β at T1, and L _ β + γ at T2 where T1 < T A and T2 > T A ,and β (0.5 B) is a congruently melting phase at a temperature higher than T B.SKETCH/SOLUTION: Given T B > T A, and T1 < T2 and T2 > T A, then at the congruent temperature, solid transforms to liquid at the same composition, hence for the threephases, α, β and γ with some solubility, is shown below along with a similar phasediagram in which there is limited solubility of B in A and A in B and the congruent phase appears as a line compound.26. FIND: Which phase diagram in Figure HP7-4 is MgO-NiO and which is NiO-CaO?Label the regions on the diagram and identify the invariant reaction.SOLUTION: To determine whether the isomorphous system is NiO-MgO or NiO-CaO, one needs to know the ionic radii of the three cations. r(Ni2+) = 0.078 nm, r(Mg2+) =0.078 nm, and r(Ca2+) = 0.106 nm. Therefore, based upon size of the ions occupying thecation sublattice we would expect the NiO-MgO system to be the isomorphous system.27. FIND: What is the maximum solid solubility of Ag in Pt?GIVEN: Phase diagram in Figure HP7-5.SOLUTION: Maximum solid solubility of Ag in Pt is 10.5 wt% Ag.28. FIND: For an alloy of peritectic composition, what is the composition of the last liquidto solidify at 1186o C?SOLUTION: The composition of the last liquid to solidify is 66.3% Ag (the peritiectic composition is 42.4% Ag).29. FIND: What is the range of alloy compositions that will peritectically transform duringequilibrium cooling?SOLUTION: Compositions between 10.5 and 66.3 wt % Ag.30. FIND: Plot the fraction of liquid, f L, the fraction of α, fα, and the fraction of β, fβ, as afunction of temperature during equilibrium cooling from 1800 to 400o C. For an alloy of peritectic composition.SOLUTION: First, you must determine the compositions of the phase(s) that is (are) in equilibrium. If, at a particular temperature the alloy is in a single phase then thecomposition of the phase is the composition of the alloy and the microstructure contains 100% of that phase. When the alloy is in a two phase field, the compositions aredetermined in using the tie-lines, and the phase fractions are determined using the lever.If, however, the microstructure is equilibrated at an invariant temperature and thecomposition lies along the invariant line, then only the composition of the phases can be determined, not their relative amounts. For an alloy of 42.4% Ag cooled underequilibrium conditions the approximate compositions of the phases in equilibrium atseveral temperatures are summarized in the following table.Fractions of phases at 1400:At 1300:At 1200At 1100At 1000At 900At 800At 700 At 600At 500At 400Fraction of phases as a function of temperature are plotted below.31. FIND: Sketch a possible diagram given the information below.GIVEN: The two component system A-B with T A > T B contains two invariant reactions of the type L + α _ β at T1, and L _ β + γ at T2 (<T1).SKETCH/SOLUTION: Reaction at T1 is a peritectic, reaction at T2 a eutectic, T A > T B and T1 > T2.In the above figure some B is soluble in A and likewise some A is soluble in B. Thephase that melts incongruently at T1 has a change in solubility with temperature.Alternatively, we can construct a diagram where the solubility of the end members are nil and the incongruently melting phase, X, is a line compound. That diagram wouldresemble the diagram shown below.32. FIND: Label all phase fields and identify the invariant reactions in the Ag-Al Phasediagram shown in figure HP7-6.SKETCH/SOLUTION:33. FIND: Label the phase fields and identify the invariant reactions in the V2O5 - NiOphase diagram.SKETCH/SOLUTION:34. FIND: Apply the 1-2-1...rule to the V2O5 - NiO diagram at 600o C.SOLUTION: At 600o C the single-phase regions are V2O5, N-V, 2N-V, 3N-V and NiO.These narrow single-phase fields define the regions of two-phase equilibrium. Also, itshould be recalled that at a fixed temperature the composition of each phase in a two-phase field is constant. All that varies is the relative amount of each phase (i.e. the lever-rule).35. FIND: Label all phase fields and identify the invariant reactions in the V2O5 - Cr2O3system.SKETCH/SOLUTION:36. FIND: For compositions X and Y, plot the fraction of phases present as a function oftemperature.SOLUTION: From the phase diagram in Figure HP7-8 the relevant we find:Composition of alloy X ~ 0.47BComposition of alloy Y ~ 0.55BComposition of the liquid at the peritectic isotherm ~ 0.44BComposition of the eutectic ~ 0.21BLiquidus temperature of X ~ 1100o CLiquidus temperature of Y ~ 1400o CPeritectic temperature ~ 900o CEutectic temperature ~ 700o CFor alloy X, in the temperature range from the liquidus (1100o C) to just above theperitectic isotherm, f L , decreases from 1.0 to:while the fraction of the oxide increases from 0 to (1 - 0.95) = 0.05. In the temperature range just below the peritectic isotherm to just above the eutectic isotherm, the phase fractions change from:andtoandjust above the eutectic temperature.In the temperature range below the eutectic isotherm, the Instal l Equa tion E ditor a nd do uble -click h ere to view equat ion.areconstant and from the lever rule are:Similarly for alloy Y:In the temperature range from the liquidus temperature (1400o C) to just above theperitectic isotherm the fraction of liquid decreases from f L = 1 towhile the fraction of Cr 2O 3 increasing from 0 toBelow the eutectic isotherm the amount of Cr 2O 3⋅V 2O 5 and Cr 2O 3 are constant.37.FIND: Identify the invariant reactions occurring in the Cu-Pb system.SOLUTION: There are two invariant reactions in the Cu - Pb system, a monotectic at 955o C and a eutectic at 326o C. For a complete illustration examine how the reduction in solubility of the components in each other effect the monotectic systems shown in Figure 7.6-1.38.FIND: Phase diagram HP7-9 indicates no mutual solid solubility of Cu in Pb or Pb in Cu. Explain why this is not strictly true.SOLUTION: Although the phase diagram appears to indicate no solubility of Cu in Pb or Pb in Cu, as pointed out on page 274 of the text, the free energy of a pure component can always be reduced by small additions of a second component since the presence of the second component increases the randomness of the system. It then is a competition between the enthalpy and the entropy terms as to how much is added, since:∆G = ∆H - T ∆S39.FIND: If an alloy containing 63 wt% Pb is cooled under equilibrium conditions from 1100o C to room temperature, plot the fraction of phases present as a function of temperature.SKETCH/SOLUTION: When an alloy containing 63% Pb, is cooled from 1100o C to room temperature under equilibrium conditions the following phases form. Below 991o C and just above the monotectic isotherm, liquid phase separation occurs. The composition of the two liquids follow the liquidus boundary, and the amount is calculated by the lever-rule. Below the monotectic isotherm to just above the eutectic isotherm two phases are present, Cu with essentially no soluble Pb and liquid rich in Pb, the composition of which follows the liquidus. Below the eutectic isotherm there is a two-phase mixture of essentially pure Cu and pure Pb. At 991o C the microstructure is essentially all liquid containing 63% Pb. At 975o C the microstructure has separated into two liquids, one (L I ) containing approximately 80% Pb and the second (L II ) containing approximately 43% Pb.Just above the monotectic isotherm.Below the monotectic isotherm, the fraction of liquid decreases, while the fraction ofsolid Cu increases. Just below 955o CAt 900o CAt 800o CThere is so little solubility of Cu in the liquid that once the temperature drops below 600o C, the system is essentially solid Cu with liquid essentially composed of Pb. Below 326o C the microstructure is a mixture of solid copper and lead. The phase diagramsuggests that there is no measurable change in solubility of the components. Then,40. FIND: Most alloying elements used in commercial titanium alloys can be classified aseither alpha stabilizers or beta stabilizers. Figure HP7.10 contains the Ti-Al and Ti-Vequilibrium phase diagrams. Which alloying element would most likely be consideredthe alpha stabilizer? Explain.GIVEN: The Ti-Al and Ti-V phase diagrams.SOLUTION: Solid titanium exists in two crystal forms. β, the high temperatureallotrope which is bcc, and α, the low temperature form which is hcp. At 882 ︒C thesetwo phases are in equilibrium at 1 atm pressure for pure titanium. When alloyingelements are added the relative stability of the phases may change. For the Ti-Al system, adding aluminum to pure titanium increases the temperature at which α begins to form.Thus, aluminum is said toIn the Ti-V system, thetemperature at which the αphases formsdecrease with increasing V,said to be a βthus V is41. FIND: In the Ti-Al system, identify a phase and its composition that melts congruently.Estimate the congruent melting temperature from the phase diagram.GIVEN: The Ti-Al phase diagram and the definition of a congruently melting phase. A phase melts congruently, when the composition of the solid and the composition of theliquid for an alloy are the same.SOLUTION: Examination of the Ti-Al phase diagram shows that the composition near5 weight % ( ~ 10 atomic %) melts congruently at approximately 1730 ︒C.42.FIND: Label all the two-phase fields on the Ti-Al phase diagram.GIVEN: The Ti-Al phase diagram with all the single phase fields labeled.SOLUTION: To complete this problem, we need only recognize that in a binary system, phases appear across the diagram alternating 1-2-1-2-...-1. Included below is the Al-Ti phase diagram with all phase fields labeled.α + Ti Al 33Ti Al + TiAl TiAl + TiAl 2α + Ti Al 3β + TiAlβ + Lα + TiAlTiAl + LTiAl + δTiAl + δ2α700900110013001500T e m p e r a t u r e , °C23TiAl + βTiAl 23TiAl + αTiAl 3αTiAl + Al 3βTiAl + Al Allotropic phaseL + δα + β3δ + βTiAl 3βTiAl + L 170043. FIND: Label all the invariant reactions occurring in the Ti-Al system.GIVEN: The Ti-Al phase diagram.SOLUTION:See the sketch of the Ti-Al diagram below.44. FIND: From the phase diagram, estimate the temperature at which Ti3Al congruentlytransforms to α Ti.GIVEN: The Ti-Al phase diagram.SOLUTION: The phase Ti3Al transforms the α Ti of the same composition atapproximately 1200︒C and a composition of approximately 21 wt % Al (33 atomic % Al).45. FIND: When the line compound β TiAl3 is heated, what is the composition of the firstliquid that forms in equilibrium with β TiAl3? Does this compound melt congruently?Explain.GIVEN: The Ti-Al phase diagram.SOLUTION: TiAl3 appears as a line compound on the phase diagram. Atapproximately 1375︒C TiAl3 undergoes a peritectic reaction on heating underequilibrium conditions. The composition of the liquid in equilibrium with TiAl3 is notthe composition of TiAl3 and hence the phase does not melt congruently, but meltsincongruently. The composition of the liquid is approximately 63 wt % Al. Theperitectic reaction written on heating is:TiAl3→ liquid (63 wt % Al) + δ Ti.46. FIND: The presence of Ti3Al in Ti-Al alloys has a detrimental effect on the ductility. Tocontrol this problem, the amount of aluminum needs to be less than 6 wt. %.Consequently, all of the products that you are producing contain a maximum of 6 wt. %of aluminum. You have been told that additions of tin, zirconium, and oxygen (oftenpresent as an impurity) are all known to be alpha stabilizers. If there is a possibility thatsmall additions of oxygen may enter the system as your alloy is melted, should youreduce the amount of aluminum in your alloy or not worry about it if ductility is animportant property for your product? Explain your reasoning.GIVEN: The Ti-Al phase diagram.SOLUTION: Since both aluminum and oxygen are α stabilizers, a judicious choicewould be to reduce the amount of aluminum in your alloy. This would be a particularlywise choice since the critical amount is just at 6 % Al, the addition of any alloyingelement that might affect the amount of Ti3Al should be avoided. Therefore reducing the amount of Al should be reduced.47. FIND: Label all the two-phase fields in the Ti-V system.GIVEN: The Ti-V phase diagram with all the single phase fields labeled.SOLUTION: To complete this problem, we need only recognize that in a binary system, phase fields appear across the diagram alternating 1-2-1-2-...-1. Included below is asketch of the Al-V phase diagram with all phase fields labeled.48. FIND: Identify the invariant reaction that is occurring at 675︒C in the Ti-V system.GIVEN: The Ti-V phase diagram.SOLUTION: The invariant reaction that is occurring at 675︒C is a monotectoid reaction and can be written symbolically as:β1→α + β2.from 900︒C down to 500︒C, plot the fraction of phases present as a function of temperature.GIVEN: The Ti-V phase diagram.SOLUTION: The compositions and the fraction of each phase from the phase diagram are shown in the table below. From the table the sketch given below illustrates the relative fraction of phases present as a function of temperature.50.FIND: Consider an alloy containing 52 wt. % V. Describe the phases present and their compositions as the alloy is cooled under equilibrium conditions from 900︒C to 500︒C. GIVEN: The Al-V phase diagram.SOLUTION: An alloy containing 52 wt. % V at 900︒C is in the β Ti,V single phase field. When the alloy is cooled it passes through the critical temperature of a monotectoid at 850︒C. Cooling below this temperature results in the formation of a two phase mixture of V-poor and V-rich solids that have the β Ti,V structure. Decreasing the temperature results in two solid phases whose compositions continuously change. The composition of the two solids follow the solid-solid miscibility boundary as shown in the sketch below. Cooling to 675︒C results in the invariant monotectoid reaction at that temperature. Symbolically that reaction can be written as β1 → α + β2. Cooling below themonotectoid temperature results in the disappearance of the β1 phase. Since the relative solubilities of V in Ti and Ti in V decrease with temperature, cooling to 500︒C changes the compositions of the α and β2 phases following the solvus boundaries as shown in the sketch.51.FIND: If a titanium alloy containing 5 wt. % V is cooled under equilibrium conditions from 900︒C down to 500︒C, plot the fraction of phases present as a function of temperature. Which phase is richer in vanadium, α or β? GIVEN: The Ti-V phase diagram.SOLUTION: The β phase is richer in vanadium than the α phase.L + β ( Ti, V )β ( Ti, V ) + α ( Ti )β1β2+Comp. of β decreases in V along phase boundary12Comp. of β increases in V along phase boundaryMonotectoid isotherm。

第1章 习题
1-10 纯铁点阵常数0.286nm ,体心立方结构,求1cm 3中有多少铁原子。
解:体心立方结构单胞拥有两个原子,单胞的体积为
V =(0.286×10-8)3 cm 3,所以1cm 3中铁原子的数目为
2283128.5510(2.8610)Fe n -=
⨯=⨯⨯
1-11 一个位错环能否各部分都是螺型位错,能否各部分都是刃型位错?为什么?
解:螺型位错的柏氏矢量与位错线平行,一根位错只有一个柏氏矢量,而一个位错环不可能与一个方向处处平行,所以一个位错环不能各部分都是螺型位错。
刃位错的柏氏矢量与位错线垂直,如果柏氏矢量垂直位错环所在的平面,则位错环处处都是刃型位错。
这种位错的滑移面是位错环与柏氏矢量方向组成的棱柱面,这种位错又称棱柱位错。
1-15 有一正方形位错线,其柏氏矢量及位错线的方向如图1-51所示。
试指出图中各段位错线的性质,并指出刃型位错额外串原子面所处的位置。
解:由柏氏矢量与位错线的关系可以知道,DC 是右螺型位错,BA 是左螺型位错。
由右手法则,CB 为正刃型位错,多余半原子面在纸面上方。
AD 为负刃型位错,多余半原子面在纸面下方。
A B
C
D。

第一章材料的结构一、解释以下基本概念空间点阵、晶格、晶胞、配位数、致密度、共价键、离子键、金属键、组元、合金、相、固溶体、中间相、间隙固溶体、置换固溶体、固溶强化、第二相强化。
二、填空题1、材料的键合方式有四类,分别是(),(),(),()。
2、金属原子的特点是最外层电子数(),且与原子核引力(),因此这些电子极容易脱离原子核的束缚而变成()。
3、我们把原子在物质内部呈()排列的固体物质称为晶体,晶体物质具有以下三个特点,分别是(),(),()。
4、三种常见的金属晶格分别为(),()和()。
5、体心立方晶格中,晶胞原子数为(),原子半径与晶格常数的关系为(),配位数是(),致密度是(),密排晶向为(),密排晶面为(),晶胞中八面体间隙个数为(),四面体间隙个数为(),具有体心立方晶格的常见金属有()。
6、面心立方晶格中,晶胞原子数为(),原子半径与晶格常数的关系为(),配位数是(),致密度是(),密排晶向为(),密排晶面为(),晶胞中八面体间隙个数为(),四面体间隙个数为(),具有面心立方晶格的常见金属有()。
7、密排六方晶格中,晶胞原子数为(),原子半径与晶格常数的关系为(),配位数是(),致密度是(),密排晶向为(),密排晶面为(),具有密排六方晶格的常见金属有()。
8、合金的相结构分为两大类,分别是()和()。
9、固溶体按照溶质原子在晶格中所占的位置分为()和(),按照固溶度分为()和(),按照溶质原子与溶剂原子相对分布分为()和()。
10、影响固溶体结构形式和溶解度的因素主要有()、()、()、()。
11、金属化合物(中间相)分为以下四类,分别是(),(),(),()。
12、金属化合物(中间相)的性能特点是:熔点()、硬度()、脆性(),因此在合金中不作为()相,而是少量存在起到第二相()作用。
13、CuZn、Cu5Zn8、Cu3Sn的电子浓度分别为(),(),()。
14、如果用M表示金属,用X表示非金属,间隙相的分子式可以写成如下四种形式,分别是(),(),(),()。


太原理工大学材料科学基础习题及参考答案第一章原子结构与结合键习题1-1计算下列粒子的德布罗意波长:(1) 质量为10-10 kg,运动速度为0.01 m·s-1的尘埃;(2) 速度为103 m/s的氢原子;(3) 能量为300 eV的自由电子。
1-2怎样理解波函数ψ的物理意义?1-3在原子结构中,ψ2和ψ2dτ代表什么?1-4写出决定原子轨道的量子数取值规定,并说明其物理意义。
1-5试绘出s、p、d轨道的二维角度分布平面图。
1-6多电子原子中,屏蔽效应和钻穿效应是怎样影响电子的能级的?1-7写出下列原子的基态电子组态(括号内为原子序号):C (6),P (15),Cl (17),Cr (24) 。
1-8 形成离子键有哪些条件?其本质是什么?1-9 试述共价键的本质。
共价键理论包括哪些理论?各有什么缺点?1-10 何谓金属键?金属的性能与金属键关系如何?1-11 范德华键与氢键有何特点和区别?参考答案:1-1 利用公式λ = h/p = h/mv 、E = hν计算德布罗意波长λ。
1-8 离子键是由电离能很小、易失去电子的金属原子与电子亲合能大的非金属原子相互作用时,产生电子得失而形成的离子固体的结合方式。
1-9 共价键是由相邻原子共有其价电子来获得稳态电子结构的结合方式。
共价键理论包括价键理论、分子轨道理论和杂化轨道理论。
1-10 当大量金属原子的价电子脱离所属原子而形成自由电子时,由金属的正离子与自由电子间的静电引力使金属原子结合起来的方式为金属建。
由于存在自由电子,金属具有高导电性和导热性;自由电子能吸收光波能量产生跃迁,表现出有金属光泽、不透明;金属正离子以球星密堆方式组成,晶体原子间可滑动,表现出有延展性。
第二章材料的结构习题2-1定义下述术语,并注意它们之间的联系和区别。
晶系,空间群,平移群,空间点阵。
2-2名词解释:晶胞与空间格子的平行六面体,并比较它们的不同点。
2-3 (1) 一晶面在x、y、z轴上的截距分别为2a、3b和6c,求出该晶面的米勒指数。
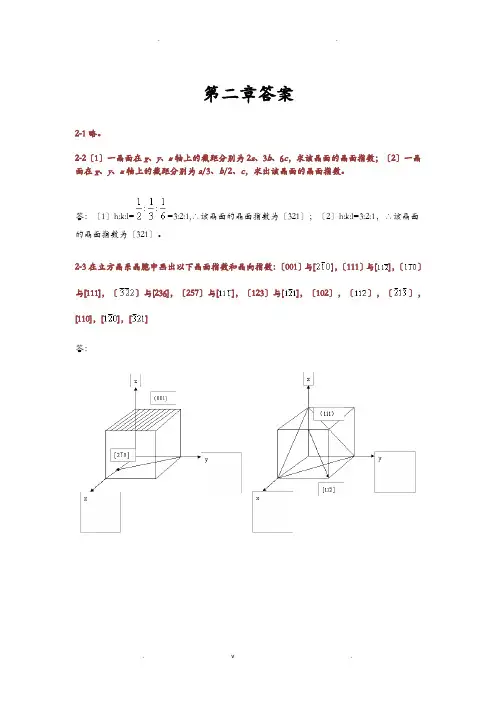
第二章答案2-1略。
2-2〔1〕一晶面在x、y、z轴上的截距分别为2a、3b、6c,求该晶面的晶面指数;〔2〕一晶面在x、y、z轴上的截距分别为a/3、b/2、c,求出该晶面的晶面指数。
答:〔1〕h:k:l==3:2:1,∴该晶面的晶面指数为〔321〕;〔2〕h:k:l=3:2:1,∴该晶面的晶面指数为〔321〕。
2-3在立方晶系晶胞中画出以下晶面指数和晶向指数:〔001〕与[],〔111〕与[],〔〕与[111],〔〕与[236],〔257〕与[],〔123〕与[],〔102〕,〔〕,〔〕,[110],[],[]答:2-4定性描述晶体构造的参量有哪些.定量描述晶体构造的参量又有哪些.答:定性:对称轴、对称中心、晶系、点阵。
定量:晶胞参数。
2-5依据结合力的本质不同,晶体中的键合作用分为哪几类.其特点是什么.答:晶体中的键合作用可分为离子键、共价键、金属键、范德华键和氢键。
离子键的特点是没有方向性和饱和性,结合力很大。
共价键的特点是具有方向性和饱和性,结合力也很大。
金属键是没有方向性和饱和性的的共价键,结合力是离子间的静电库仑力。
范德华键是通过分子力而产生的键合,分子力很弱。
氢键是两个电负性较大的原子相结合形成的键,具有饱和性。
2-6等径球最严密堆积的空隙有哪两种.一个球的周围有多少个四面体空隙、多少个八面体空隙.答:等径球最严密堆积有六方和面心立方严密堆积两种,一个球的周围有8个四面体空隙、6个八面体空隙。
2-7n个等径球作最严密堆积时可形成多少个四面体空隙、多少个八面体空隙.不等径球是如何进展堆积的.答:n个等径球作最严密堆积时可形成n个八面体空隙、2n个四面体空隙。
不等径球体进展严密堆积时,可以看成由大球按等径球体严密堆积后,小球按其大小分别填充到其空隙中,稍大的小球填充八面体空隙,稍小的小球填充四面体空隙,形成不等径球体严密堆积。
2-8写出面心立方格子的单位平行六面体上所有结点的坐标。
答:面心立方格子的单位平行六面体上所有结点为:〔000〕、〔001〕〔100〕〔101〕〔110〕〔010〕〔011〕〔111〕〔0〕〔0〕〔0〕〔1〕〔1〕〔1〕。

第一章材料的结构一、解释以下基本概念空间点阵、晶格、晶胞、配位数、致密度、共价键、离子键、金属键、组元、合金、相、固溶体、中间相、间隙固溶体、置换固溶体、固溶强化、第二相强化。
二、填空题1、材料的键合方式有四类,分别是(),(),(),()。
2、金属原子的特点是最外层电子数(),且与原子核引力(),因此这些电子极容易脱离原子核的束缚而变成()。
3、我们把原子在物质内部呈()排列的固体物质称为晶体,晶体物质具有以下三个特点,分别是(),(),()。
4、三种常见的金属晶格分别为(),()和()。
5、体心立方晶格中,晶胞原子数为(),原子半径与晶格常数的关系为(),配位数是(),致密度是(),密排晶向为(),密排晶面为(),晶胞中八面体间隙个数为(),四面体间隙个数为(),具有体心立方晶格的常见金属有()。
6、面心立方晶格中,晶胞原子数为(),原子半径与晶格常数的关系为(),配位数是(),致密度是(),密排晶向为(),密排晶面为(),晶胞中八面体间隙个数为(),四面体间隙个数为(),具有面心立方晶格的常见金属有()。
7、密排六方晶格中,晶胞原子数为(),原子半径与晶格常数的关系为(),配位数是(),致密度是(),密排晶向为(),密排晶面为(),具有密排六方晶格的常见金属有()。
8、合金的相结构分为两大类,分别是()和()。
9、固溶体按照溶质原子在晶格中所占的位置分为()和(),按照固溶度分为()和(),按照溶质原子与溶剂原子相对分布分为()和()。
10、影响固溶体结构形式和溶解度的因素主要有()、()、()、()。
11、金属化合物(中间相)分为以下四类,分别是(),(),(),()。
12、金属化合物(中间相)的性能特点是:熔点()、硬度()、脆性(),因此在合金中不作为()相,而是少量存在起到第二相()作用。
13、CuZn、Cu5Zn8、Cu3Sn的电子浓度分别为(),(),()。
14、如果用M表示金属,用X表示非金属,间隙相的分子式可以写成如下四种形式,分别是(),(),(),()。

材料科学基础答案(精⼼整理)第1章晶体结构1.在⽴⽅晶系中,⼀晶⾯在x轴的截距为1,在y轴的截距为1/2,且平⾏于z 轴,⼀晶向上某点坐标为x=1/2,y=0,z=1,求出其晶⾯指数和晶向指数,并绘图⽰之。
2.画出⽴⽅晶系中下列晶⾯和晶向:(010),(011),(111),(231),(321),[010], [011],[111],[231],[321]。
3.纯铝晶体为⾯⼼⽴⽅点阵,已知铝的相对原⼦质量Ar(Al)=27,原⼦半径r=0.143nm,求铝晶体的密度。
4.何谓晶体?晶体与⾮晶体有何区别?5.试举例说明:晶体结构与空间点阵?单位空间格⼦与空间点阵的关系?6.什么叫离⼦极化?极化对晶体结构有什么影响?7.何谓配位数(离⼦晶体/单质)?8.何谓对称操作,对称要素?9.计算⾯⼼⽴⽅结构(111)与(100)晶⾯的⾯间距及原⼦密度(原⼦个数/单位⾯积)。
10.已知室温下α-Fe(体⼼)的点阵常数为0.286nm,分别求(100)、(110)、(123)的晶⾯间距。
11.已知室温下γ-Fe(⾯⼼)的点阵常数为0.365nm,分别求(100)、(110)、(112)的晶⾯间距。
12.已知Cs+半径为0.170nm,Cl-半径为0.181 nm,计算堆积系数。
13.MgO 属NaCl型结构,若rMg 2+=0.078nm,rO2-=0.132nm,(1)试⽤鲍林规则分析氧化镁晶体结构?(2)计算堆积密度?(3)画出氧化镁在(100)、(110)、(111)晶⾯上的结点和离⼦排布图?答案1.答:晶⾯指数为:(120),见图ABCD ⾯;晶向指数为:[102],见图OP 向。
2.答:3. 4. 5.6. 答:离⼦极化:在离⼦紧密堆积时,带电荷的离⼦所产⽣的电场必然要对另⼀离⼦的电⼦云发⽣作⽤(吸引或排斥),因⽽使这个离⼦的⼤⼩和形状发⽣了改变,这种现象叫离⼦极化。
极化会对晶体结构产⽣显著影响,主要表现为极化会导致离⼦间距离缩短,离⼦配位数降低,同时变形的电⼦云相互重叠,使键性由离⼦键向共价键过渡,最终使晶体结构类型发⽣变化。

材料科学基础课后习题答案材料科学基础课后习题答案第一章:晶体结构和晶体缺陷1. 什么是晶体?晶体的特点是什么?答:晶体是由有序排列的原子、离子或分子组成的固态材料。
晶体的特点包括有规则的、重复的、周期性的结构,具有明确的晶体面和晶面间角度。
2. 简述晶体中离子束缚以及普通共价键束缚的区别?答:晶体中离子束缚是指由电荷相反的离子通过电磁力相互吸引而形成的结合力,例如NaCl晶体。
普通共价键束缚是由共享电子对形成的,例如金刚石晶体。
离子束缚通常较为强烈,晶体具有高熔点和脆性;而共价键束缚相对较弱,晶体具有低熔点和韧性。
3. 什么是晶体缺陷?列举几种晶体缺陷并简要描述其影响。
答:晶体缺陷是指晶体中排列异常的原子、离子或分子。
常见的晶体缺陷包括点缺陷、线缺陷和面缺陷。
点缺陷指的是晶体中原子位置的缺失或替代,如空位、间隙原子和杂质原子;线缺陷是晶体中晶面上原子位置的错误,如位错和螺旋位错;面缺陷是指晶面之间的错配,如晶界和孪生界。
这些晶体缺陷会影响晶体的物理性质和力学性能。
4. 什么是晶体结构中的定义因素?它们的作用是什么?答:晶体结构中的定义因素包括晶胞和晶格参数。
晶胞是最小重复单元,由一定数量的晶体中的原子、离子或分子组成。
晶格参数描述晶胞的大小和形状。
晶胞和晶格参数共同定义了晶体的结构。
晶胞和晶格参数的作用是确定晶体的晶体面、晶面间角度以及晶体的物理性质。
5. 什么是晶格点?晶格点的种类有哪些?答:晶格点是位于晶体内部的原子、离子或分子的位置。
晶格点的种类包括普通晶格点、间隙晶格点和特殊晶格点。
普通晶格点是晶体中原子、离子或分子的晶格点,如AB型晶体中的A和B原子;间隙晶格点是晶体中没有原子、离子或分子的晶格点,如金刚石中的间隙晶格点;特殊晶格点是具有非普通晶格点性质的晶体中的晶格点,如晶体中的空位或杂质原子。
第二章:物质的结构与性能关系1. 简述晶体结构对物质性能的影响。
答:晶体结构直接影响物质的物理性质和化学性质。
《材料科学基础》课后习题答案第一章材料结构的基本知识4. 简述一次键和二次键区别答:根据结合力的强弱可把结合键分成一次键和二次键两大类。
其中一次键的结合力较强,包括离子键、共价键和金属键。
一次键的三种结合方式都是依靠外壳层电子转移或共享以形成稳定的电子壳层,从而使原子间相互结合起来。
二次键的结合力较弱,包括范德瓦耳斯键和氢键。
二次键是一种在原子和分子之间,由诱导或永久电偶相互作用而产生的一种副键。
6. 为什么金属键结合的固体材料的密度比离子键或共价键固体为高?答:材料的密度与结合键类型有关。
一般金属键结合的固体材料的高密度有两个原因:(1)金属元素有较高的相对原子质量;(2)金属键的结合方式没有方向性,因此金属原子总是趋于密集排列。
相反,对于离子键或共价键结合的材料,原子排列不可能很致密。
共价键结合时,相邻原子的个数要受到共价键数目的限制;离子键结合时,则要满足正、负离子间电荷平衡的要求,它们的相邻原子数都不如金属多,因此离子键或共价键结合的材料密度较低。
9. 什么是单相组织?什么是两相组织?以它们为例说明显微组织的含义以及显微组织对性能的影响。
答:单相组织,顾名思义是具有单一相的组织。
即所有晶粒的化学组成相同,晶体结构也相同。
两相组织是指具有两相的组织。
单相组织特征的主要有晶粒尺寸及形状。
晶粒尺寸对材料性能有重要的影响,细化晶粒可以明显地提高材料的强度,改善材料的塑性和韧性。
单相组织中,根据各方向生长条件的不同,会生成等轴晶和柱状晶。
等轴晶的材料各方向上性能接近,而柱状晶则在各个方向上表现出性能的差异。
对于两相组织,如果两个相的晶粒尺度相当,两者均匀地交替分布,此时合金的力学性能取决于两个相或者两种相或两种组织组成物的相对量及各自的性能。
如果两个相的晶粒尺度相差甚远,其中尺寸较细的相以球状、点状、片状或针状等形态弥散地分布于另一相晶粒的基体内。
如果弥散相的硬度明显高于基体相,则将显著提高材料的强度,同时降低材料的塑韧性。
武汉理工大学材料科学基础(第2版)课后习题和答案第一章绪论1、仔细观察一下白炽灯泡,会发现有多少种不同的材料?每种材料需要何种热学、电学性质?2、为什么金属具有良好的导电性和导热性?3、为什么陶瓷、聚合物通常是绝缘体?4、铝原子的质量是多少?若铝的密度为2.7g/cm3,计算1mm3中有多少原子?5、为了防止碰撞造成纽折,汽车的挡板可有装甲制造,但实际应用中为何不如此设计?说出至少三种理由。
6、描述不同材料常用的加工方法。
7、叙述金属材料的类型及其分类依据。
8、试将下列材料按金属、陶瓷、聚合物或复合材料进行分类:黄铜钢筋混凝土橡胶氯化钠铅-锡焊料沥青环氧树脂镁合金碳化硅混凝土石墨玻璃钢9、Al2O3陶瓷既牢固又坚硬且耐磨,为什么不用Al2O3制造铁锤?第二章晶体结构1、解释下列概念晶系、晶胞、晶胞参数、空间点阵、米勒指数(晶面指数)、离子晶体的晶格能、原子半径与离子半径、配位数、离子极化、同质多晶与类质同晶、正尖晶石与反正尖晶石、反萤石结构、铁电效应、压电效应.2、(1)一晶面在某、y、z轴上的截距分别为2a、3b、6c,求出该晶面的米勒指数;(2)一晶面在某、y、z轴上的截距分别为a/3、b/2、c,求出该晶面的米勒指数。
3、在立方晶系的晶胞中画出下列米勒指数的晶面和晶向:(001)与[210],(111)与[112],(110)与[111],(322)与[236],(257)与[111],(123)与[121],(102),(112),(213),[110],[111],[120],[321]4、写出面心立方格子的单位平行六面体上所有结点的坐标。
5、已知Mg2+半径为0.072nm,O2-半径为0.140nm,计算MgO晶体结构的堆积系数与密度。
6、计算体心立方、面心立方、密排六方晶胞中的原子数、配位数、堆积系数。
7、从理论计算公式计算NaC1与MgO的晶格能。
MgO的熔点为2800℃,NaC1为80l℃,请说明这种差别的原因。
第一章 原子排列与晶体结构1. [110], (111), ABCABC…, 0.74 , 12 , 4 ,a r 42=; [111], (110) , 0.68 , 8 , 2 , a r 43= ; ]0211[, (0001) , ABAB , 0.74 , 12 , 6 , 2a r =。
2.0.01659nm 3 , 4 , 8 。
3.FCC , BCC ,减少 ,降低 ,膨胀 ,收缩 。
4. 解答:见图1-1 5. 解答:设所决定的晶面为(hkl ),晶面指数与面上的直线[uvw]之间有hu+kv+lw=0,故有:h+k-l=0,2h-l=0。
可以求得(hkl )=(112)。
6 解答:Pb 为fcc 结构,原子半径R 与点阵常数a 的关系为a r 42=,故可求得a =0.4949×10-6mm 。
则(100)平面的面积S =a 2=0.244926011×0-12mm 2,每个(100)面上的原子个数为2。
所以1 mm 2上的原子个数s n 1==4.08×1012。
第二章 合金相结构一、 填空1) 提高,降低,变差,变大。
2) (1)晶体结构;(2)元素之间电负性差;(3)电子浓度 ;(4)元素之间尺寸差别3) 存在溶质原子偏聚 和短程有序 。
4) 置换固溶体 和间隙固溶体 。
5) 提高 ,降低 ,降低 。
6) 溶质原子溶入点阵原子溶入溶剂点阵间隙中形成的固溶体,非金属原子与金属原子半径的比值大于0.59时形成的复杂结构的化合物。
二、 问答1、 解答: α-Fe 为bcc 结构,致密度虽然较小,但是它的间隙数目多且分散,间隙半径很小,四面体间隙半径为0.291Ra ,即R =0.0361nm ,八面体间隙半径为0.154Ra ,即R =0.0191nm 。
氢,氮,碳,硼由于与α-Fe 的尺寸差别较大,在α-Fe 中形成间隙固溶体,固溶度很小。
材料科学基础习题答案《材料科学基础》习题参考答案第一章原子结构与键合★考前复习范围概念:4个量子数、3个准则、金属键、离子键、共价键1.原子中一个电子的空间位置和能量可用哪四个量子数来决定?在多电子的原子中,核外电子的排布应遵循哪些原则?答:1).主量子数n=1、2、3、4(K、L、M、N)决定原子中电子能量以及与核的平均距离,即电子所处的量子壳层。
2).轨道角量子数li=0~(n-1),(s,p,d,f,g)给出电子在同一量子壳层内所处的能级。
(亚层)3).磁量子数mi,给出每个轨道角动量量子数的轨道数或能级数,每个li下的磁量子总数为2li+1。
(能级)4).自旋角量子数si=±1/2, 反映电子不同的自旋方向。
(电子数)Pauli不相容原理:在同一个原子中没有四个量子数完全相同的电子。
能量最低原理:电子在原子中所处的状态,总是尽可能分布到能量最低的轨道上。
Hund规则:电子分布到能量相同的等价轨道上时,总是尽先以自旋相同的方向,单独占据能量相同的轨道。
2.在元素周期表中,同一周期或同一主族元素原子结构有什么共同特点?从左到右或从上到下元素结构有什么区别?性质如何递变?答:同一周期元素具有相同原子核外电子层数,但从左到右,核电荷依次增多,原子半径逐渐减小,电离能增加,失电子能力降低,得电子能力增加,金属性减弱,非金属性增强;同一主族元素核外电子数相同,但从上到下,电子层数增多,原子半径增大,电离能降低,失电子能力增加,得电子能力降低,金属性增加,非金属性降低。
3.何谓同位素?为什么元素的相对原子质量不总为正整数?答:在元素周期表中占据同一位置,尽管它们的质量不同,然它们的化学性质相同的物质称为同位素。
由于各同位素的含中子量不同(质子数相同),故具有不同含量同位素的元素总的相对原子质量不为正整数。
4.铬的原子序数为24,它共有四种同位素:4.31%的Cr原子含有26个中子,83.76%含28个中子,9.55%含有29个中子,且2.38%含有30个中子。
第一章8.计算下列晶体的离于键与共价键的相对比例(1)NaF (2)CaO (3)ZnS解:1、查表得:X Na =0.93,X F =3.98根据鲍林公式可得NaF 中离子键比例为:21(0.93 3.98)4[1]100%90.2%e ---⨯=共价键比例为:1-90.2%=9.8% 2、同理,CaO 中离子键比例为:21(1.00 3.44)4[1]100%77.4%e---⨯=共价键比例为:1-77.4%=22.6%3、ZnS 中离子键比例为:21/4(2.581.65)[1]100%19.44%ZnS e --=-⨯=中离子键含量共价键比例为:1-19.44%=80.56%10说明结构转变的热力学条件与动力学条件的意义.说明稳态结构与亚稳态结构之间的关系。
答:结构转变的热力学条件决定转变是否可行,是结构转变的推动力,是转变的必要条件;动力学条件决定转变速度的大小,反映转变过程中阻力的大小。
稳态结构与亚稳态结构之间的关系:两种状态都是物质存在的状态,材料得到的结构是稳态或亚稳态,取决于转交过程的推动力和阻力(即热力学条件和动力学条件),阻力小时得到稳态结构,阻力很大时则得到亚稳态结构。
稳态结构能量最低,热力学上最稳定,亚稳态结构能量高,热力学上不稳定,但向稳定结构转变速度慢,能保持相对稳定甚至长期存在。
但在一定条件下,亚稳态结构向稳态结构转变。
第二章1.回答下列问题:(1)在立方晶系的晶胞内画出具有下列密勒指数的晶面和晶向:(001)与[210],(111)与[112],(110)与 [111],(132)与[123],(322)与[236](2)在立方晶系的一个晶胞中画出(111)和 (112)晶面,并写出两晶面交线的晶向指数。
(3)在立方晶系的一个晶胞中画出同时位于(101). (011)和(112)晶面上的[111]晶向。
解:1、2.有一正交点阵的 a=b, c=a/2。
某晶面在三个晶轴上的截距分别为 6个、2个和4个原子间距,求该晶面的密勒指数。
3.立方晶系的 {111}, 1110}, {123)晶面族各包括多少晶面?写出它们的密勒指数。
4.写出六方晶系的{1012}晶面族中所有晶面的密勒指数,在六方晶胞中画出[1120]、[1101]晶向和(1012)晶面,并确定(1012)晶面与六方晶胞交线的晶向指数。
5.根据刚性球模型回答下列问题:(1)以点阵常数为单位,计算体心立方、面心立方和密排六方晶体中的原子半径及四面体和八面体的间隙半径。
(2)计算体心立方、面心立方和密排六方晶胞中的原子数、致密度和配位数。
6.用密勒指数表示出体心立方、面心立方和密排六方结构中的原子密排面和原子密排方向,并分别计算这些晶面和晶向上的原子密度。
解:1、体心立方密排面:{110}2114 1.414a -+⨯= 密排方向:<111> 11.15a -= 2、面心立方密排面:{111}21133 2.3a -⨯+⨯=密排方向:<110>11.414a -= 3、密排六方密排面:{0001}2161 1.15a -⨯+= 密排方向:1120<>,原子密度:122a a-= 7.求下列晶面的晶面间距,并指出晶面间距最大的晶面 :(1)已知室温下 α-Fe 的点阵常数为0. 286nm ,分别求出 (100), (110), (123)的晶面间距 。
(2)已知 9160C 时γ-Fe 的点阵常数为 0. 365nm ,分别求出 (100), (111), (112)的晶面间距 。
(3)已知室温下 Mg 的点阵常数为a=0.321nm, c=0. 521nm ,分别求出 (1120), (1010),(1012)的晶面间距。
8.回答下列问题:(1)通过计算判断(110), (132), (311)晶面是否属于同一晶带?(2)求 (211)和 ((110)晶面的晶带轴 ,并列出五个属于该晶带的晶面的密勒指数。
解:1、根据晶带定律,hu+kv+lw=0,可得(110), (132)的晶带轴为[112] 3×1+1×1-2×1=2≠0或 (132), (311)的晶带轴为[158] -1×1+1×5-0×8=4≠0故(110), (132), (311)晶面不属于同一晶带 2、根据晶带定律,hu+kv+lw=0,可得 2u+v+w=0 u+v=0联立求解,得:u:v:w=-1:1:1,故晶带轴为[111] 属于该晶带的晶面:(321)、(312)、(101)、(01)、(431) 等。
9.回答下列问题:(1)试求出立方晶系中[321]与[401]晶向之间的夹角。
(2)试求出立方晶系中(210)与(320)晶面之间的夹角。
(3)试求出立方晶系中(111)晶面与[112]晶向之间的夹角。
解:1、根据晶向指数标定法可知:矢量32OA i j k =++必然平行于[321]晶向 矢量4OB i k =+必然平行于[401]晶向则:这两个矢量夹角即为[321]与[401]晶向之间的夹角 根据矢量点积公式:cosOA OB OA OB α= 即13α= 或2AB OB OA i k =-=-矢量,,OA OBAB根据余弦定理:51417α=+- 解得: α=32.58°2、立方系中同指数的晶面与晶向相互垂直,故(210)与(320)晶面之间的夹角与[210]与[320]晶向之间的夹角相等, 根据晶向指数标定法可知:矢量21OA i j =+必然平行于[210]晶向 矢量32OB i j =+必然平行于[320]晶向则:这两个矢量夹角即为[210]与[320]晶向之间的夹角 根据矢量点积公式:cosOA OB OA OB α= 即8α=或AB OB OA i j =-=+矢量,,OA OB AB根据余弦定理:2513α=+- 解得: α=7.1°3、由于(111)晶面与[112]晶向之间满足晶带定律:hu+kv+lw=0,根据晶带定律可知,立方晶系中(111)晶面与[112]晶向平行,故他们之间的夹角为0°。
方法2,1、求[111]与[112]之间夹角为90° 2、(111)与[112]之间夹角为0°第四章1.纯 Cu 的空位形成能为 1.5aJ/atom (1aJ=10-18J),将纯Cu 加热至850℃后激冷至室温 (20℃),若高温下的空位全部保留,试求过饱和空位浓度与室温平衡空位浓度的比值。
解:平衡空位浓度:expv uC A kT-= 8508502085020201823274.2exp11exp[()]exp1.51011exp[()]1.3810850273.1520273.15uA C u kT u C k T T A kT e ----==---⨯=-⨯++= 2.已知银在 800℃下的平衡空位数为 3. 6 ×1023/m 3,该温度下银的密度ρAg = 9. 58g/crn 3 ,银的摩尔质量为M Ag =107.9g/mol ,计算银的空位形成能。
解:平衡空位浓度:exp v uC A kT-= 1m 3内银原子总数:23602836.02109.5810 5.3410/107.9Ag AgN N m M ρ⨯⨯⨯===⨯2328233.6101exp5.3410 1.3810(800273.15)u-⨯-=⨯⨯⨯+ 191.7610/u J atom -=⨯3.空位对材料行为的主要影响是什么?4.某晶体中有一条柏氏矢量为a [001]的位错线,位错线的一端露头于晶体表面,另一端与两条位错线相连接,其中一条的柏氏矢量为/2[111]a ,求另一条位错线的柏氏矢量。
答:根据柏氏矢量的守恒性,另一条位错的柏氏矢量为:[110][111][111]22a aa -=5.在图 4-52所示的晶体中,ABCD 滑移面上有一个位错环,其柏氏矢量b 平行于AC(1)指出位错环各部分的位错类型。
(2)在图中表示出使位错环向外运动所需施加的切应力方向。
(3)该位错环运动出晶体后,晶体外形如何变化?答:(1)位错环和与AC 平行的直线相切的部分为纯螺位错,位错环和与 AC 垂直的直线相切的部分为纯刃位错,其余部分为混合位错,作图(2)切应力与b 平行,作用在晶体上下两面上。
t ×b →多余原子面,作图 (3)沿b 方向滑出一个柏氏矢量单位的距离6.在图 4-53所示的晶体中有一位错线 fed, de 段正好处于位错的滑移面上,of 段处于非滑移面上,位错的柏氏矢量 b 与 AB 平行而垂直于 BC, (1)欲使de 段位错线在 ABCD 滑移面上运动 (of 段因处于非滑移面是固定不动的),应对晶体施加怎样的应力? (2)在 上述 应力作用下 de 段位错线如何运动 ,晶体外协 如 1可贾 化 ?7.在图 4-54所示的面心立方 晶体 的 (111)滑移 面上有 两条弯 折 的位错线 OS 和 O'S'其 中 O'S'位错 的台阶垂直十 ((111),它们的桕氏天量如图中箭头P)rT o 0)判断位错线上各段位错的类型。
(2)有一切应力施加于滑移面,且与柏氏矢量平行时,两条位错线的滑移特征有何差异?8.在两个相互垂直的滑移面上各有一条刃型位错线,位错线的柏氏矢量如图 4-55a, b 所示。
设其中一条位错线 AB 在切应力作用下发生如图所示的运动,试问交截后两条位错线的形状有何变化?各段位错线的位错类型是什么? (1)交截前两条刃位错的柏氏矢量相互垂直的情况 (图a) (2)交截前两条刃位错的柏氏矢量相互平行的情况 (图b) 9.在晶体的同一滑移面上有两个直径分别为 r ;和r :的位错环,其中rl>r2,它们的柏氏矢量相同,试问在切应力作用下何者更容易运动?为什么? 10.判断下列位错反应能否进行:[101][121][111]263a a a+→ 几何条件:[101][121][222][111]2663a a a a+==能量条件:反应前 222223b a =+=∑反应后22213b a ==∑ 满足几何条件和能量条件,故反应能够进行。
[100][101][101]22a aa →+ 几何条件:[101][101][200][100]222a a aa +==能量条件:反应前222(b a ==∑反应后222b a ==∑满足几何条件,但反应前后能量相等,不满足能量条件,故无外力作用时,该位错反应不能进行。
[112][111][111]362a a a+→ 几何条件:[112][111][333][111]3662a a a a+==能量条件:反应前 222234b a =+=∑反应后22234b a ==∑ 满足几何条件,但反应前后能量相等,不满足能量条件,故无外力作用时,该位错反应不能进行。