
Preparation of n -bromobutane
一、Purpose
1、Study the principle and method of preparing n-butyl bromide from n-butyl alcohol by treatment with sodium bromide and concentrated sulfuric acid
2、Learn the technique of reflux with a gas trap apparatus and washing.
二、Principle
n-Butyl bromide can be easily prepared by allowing n-butyl alcohto react with sodium bromide and concentrated sulfuric acid.Main reactions :
NaBr + H 2SO4 → HBr + NaHSO4
24
H SO 322232222CH CH CH CH OH HBr CH CH CH CH Br H O
+???→+Secondary reactions :
;
24H SO 32223222CH CH CH CH OH CH CH CH=CH H O
???→+()24H SO 32223222222CH CH CH CH OH CH CH CH CH O H O
???→+24222H SO HBr Br SO H O
+??→++三、Materials n-butyl alcohol :4mL Sodium bromide :5g
Concentrated sulfuric acid :2.5mL/6mL Anhydrous calcium chloride:0.5g 10% aqueous sodium hydroxide:5mL
四、Primary reagent And Product physical constants
Name
Relative molecular mass
Character
Relative density Melting point Boiling point Refractive index
n -bromobutane
137.03colorless and transparent liquid 1.299-122.4101.6 1.4399n-butyl alcohol
74.12
colorless and transparent liquid
0.8098
-89.2
117.7
1.3993
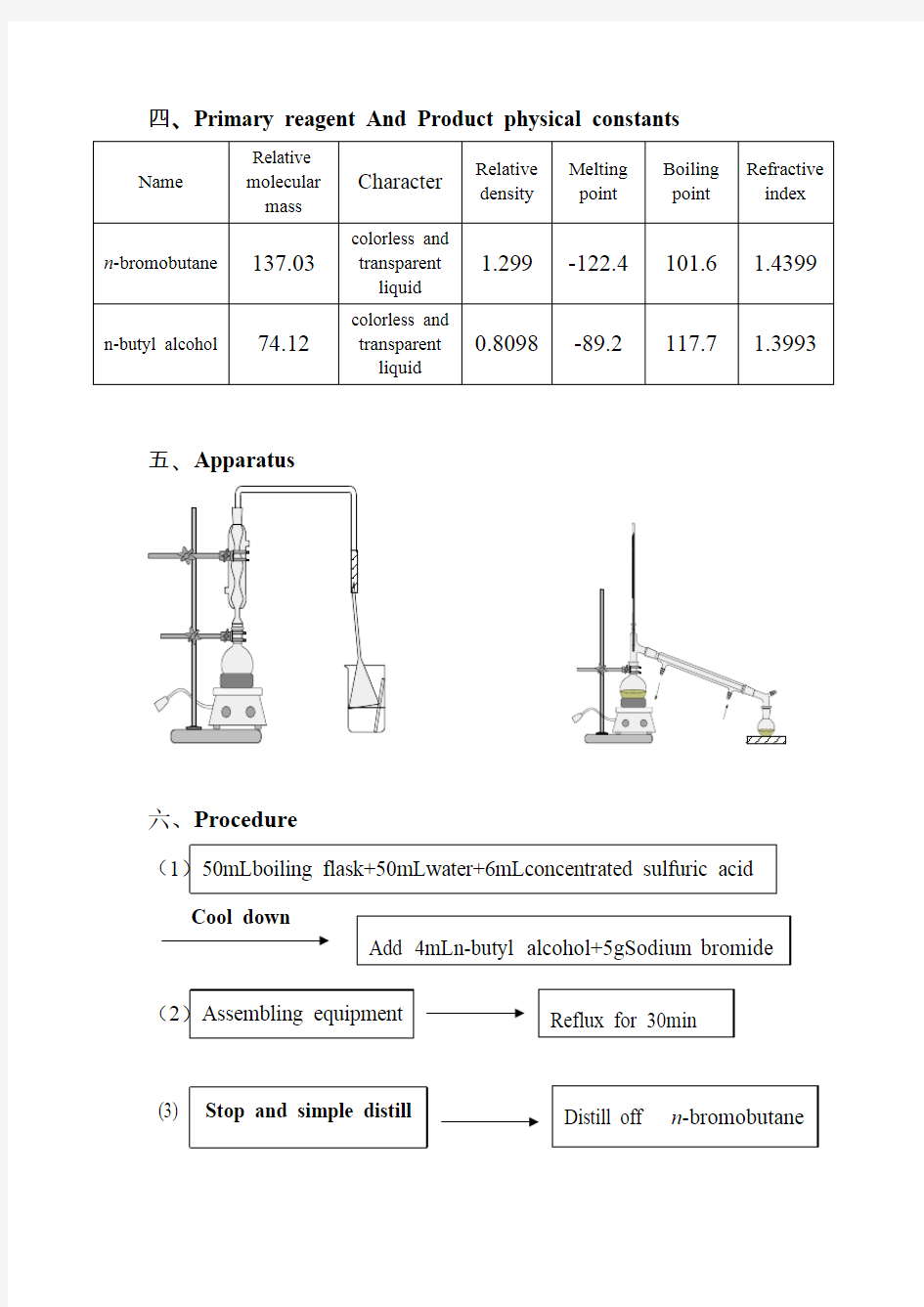
五、Apparatus
六、Procedure
(1)50mLboiling flask+50mLwater+6mLconcentrated sulfuric acid
Cool down
(2)Assembling equipment
(3) Stop and simple distill
(4)
(5)
七、Experimental records
(1) Sulfuric acid soluble in water gives off a lot of heat
(2) The solution of the distillation flask become yellow and the sodium bromide dissolve
(3)Solution is divided into two layers and liquid of the distillation become clear
(4) Liquid layer, upper as the water phase, the lower is positive bromobutane and liquid for the milky haze
(5) Liquid at 99 ℃ and stable distillation, after rising to 103 ℃, 103 ℃after fractions and the former part of the don't mix.
八、Data recording
Output:1.3g theoretical yield:5.8g productivity:21.7% Character: colorless and transparent liquid Refractive index:1.4372
九、Experiment Discussion
1、Turbidity is because it contains a variety of organic phase to organic
impurities
2、Plus the bottle stopper of calcium chloride anhydrous dry battery in
order to prevent the water vapor in the air into the conical flask, at the same time prevent product turbidity