Isolated Star Formation A Compact HII Region in the Virgo Cluster
- 格式:pdf
- 大小:115.73 KB
- 文档页数:5

海岛棉DELLA蛋⽩编码基因GbGAI的克隆与功能初步分析0引⾔海岛棉(长绒棉)(Gossiypium barbadense L.)的纤维具有长、细、强等突出特点,在棉花栽培种中品质最优。
长绒棉⽣产对于提⾼中国纺织产品档次、增强⾏业竞争⼒发挥了重要作⽤[1]。
20世纪90年代以后,中国选育出了纤维品质性状相对优异的海岛棉新品种,如新海l3号、新海l5号、新海l7号等。
这些品种的主要纤维物理指标,已经能够与世界上品质最优的超级长绒棉品种相媲美,只是麦克隆值偏⼤,对纺优质⾼档纱来说纤维较粗。
因此,中国选育的长绒棉品种的综合品质性状尚需进⼀步改良[2]。
⾚霉素(gibberellin ,GAs )是⼀类重要的植物激基⾦项⽬:国家⾃然科学基⾦(307600991);兵团博⼠资⾦(2007JC08);⽯河⼦⼤学博⼠基⾦(RCZ200620)。
第⼀作者简介:曲廷云,男,1984年出⽣,⼭东德州⼈,在读硕⼠⽣,主要从事植物基因⼯程研究。
E-mail :qty1385@/doc/b62940039.html。
通讯作者:郑银英,⼥,1975年出⽣,硕⼠⽣导师,博⼠,从事植物⽣理⽣化与分⼦⽣物学研究。
通信地址:832003新疆⽯河⼦市北四路⽯河⼦⼤学农业⽣物技术重点实验室,E-mail :zhengyinying@/doc/b62940039.html。
收稿⽇期:2010-02-04,修回⽇期:2010-03-08。
海岛棉DELLA 蛋⽩编码基因GbGAI 的克隆与功能初步分析曲廷云1,王拴锁2,彭明1,3,崔百明1,郑银英1(1⽯河⼦⼤学农业⽣物技术重点实验室⁄⽯河⼦⼤学⽣命科学院,新疆⽯河⼦832003;2中国科学院遗传与发育⽣物学研究所植物细胞与染⾊体⼯程国家重点实验室,北京100101;3中国热带农业科学院热带⽣物技术研究所,海⼝571101)摘要:⾚霉素在棉花纤维发育过程中起着重要作⽤。
DELLA 蛋⽩是⾚霉素信号传导通路中的⼀类重要负调节因⼦。

ARTICLEOPENReceived8Apr2015|Accepted25Aug2015|Published29Sep2015Entropy-stabilized oxidesChristina M.Rost1,Edward Sachet1,Trent Borman1,Ali Moballegh1,Elizabeth C.Dickey1,Dong Hou1,Jacob L.Jones1,Stefano Curtarolo2&Jon-Paul Maria1Configurational disorder can be compositionally engineered into mixed oxide by populating asingle sublattice with many distinct cations.The formulations promote novel and entropy-stabilized forms of crystalline matter where metal cations are incorporated in new ways.Here,through rigorous experiments,a simple thermodynamic model,and afive-componentoxide formulation,we demonstrate beyond reasonable doubt that entropy predominatesthe thermodynamic landscape,and drives a reversible solid-state transformation betweena multiphase and single-phase state.In the latter,cation distributions are proven tobe random and homogeneous.Thefindings validate the hypothesis that deliberateconfigurational disorder provides an orthogonal strategy to imagine and discover new phasesof crystalline matter and untapped opportunities for property engineering.1Department of Materials Science and Engineering,North Carolina State University,Raleigh,North Carolina27695,USA.2Department of Mechanical Engineering and Materials Science,Center for Materials Genomics,Duke University,Durham,North Carolina27708,USA.Correspondence and requests for materials should be addressed to J.-P.M.(email:jpmaria@)or to S.C.(email:stefano@).A grand challenge facing materials science is the continuoushunt for advanced materials with properties that satisfythe demands of rapidly evolving technology needs.The materials research community has been addressing this problem since the early1900s when Goldschmidt reported the‘the method of chemical substitution’1that combined a tabulation of cationic and anionic radii with geometric principles of ion packing and ion radius ratios.Despite its simplicity,this model enabled a surprising capability to predict stable phases and structures.As early as1926many of the technologically important materials that remain subjects of contemporary research were identified (though their properties were not known);BaTiO3,AlN,GaP, ZnO and GaAs are among that list.These methods are based on overarching natural tendencies for binary,ternary and quaternary structures to minimize polyhedral distortions,maximize spacefilling and adopt polyhedral linkages that preserve electroneutrality1–3.The structure-field maps compiled by Muller and Roy catalogue the crystallographic diversity in the context of these largely geometry-based predictions4.There are,however,limitations to the predictive power,particularly when factors like partial covalency and heterodesmic bonding are considered.To further expand the library of advanced materials and property opportunities,our community explores possibilities based on mechanical strain5,artificial layering6,external fields7,combinatorial screening8,interface engineering9,10and structuring at the nanoscale6,11.In many of these efforts, computation and experiment are important companions.Most recently,high-throughput methods emerged as a power-ful engine to assess huge sections of composition space12–17and identified rapidly new Heusler alloys,extensive ion substitution schemes18,19,new18-electron ABX compounds20and new ferroic semiconductors21.While these methods offer tremendous predictive power and an assessment of composition space intractable to experiment, they often utilize density functional theory calculations conducted at0K.Consequently,the predicted stabilities are based on enthalpies of formation.As such,there remains a potential section of discovery space at elevated temperatures where entropy predominates the free-energy landscape.This landscape was explored recently by incorporating deliberatelyfive or more elemental species into a single lattice with random occupancy.In such crystals,entropic contributions to the free energy,rather than the cohesive energy, promote thermodynamic stability atfinite temperatures.The approach is being explored within the high-entropy-alloy family of materials(HEAs)22,in which extremely attractive properties continue to be found23,24.In HEAs,however,discussion remains regarding the true role of configurational entropy25–28, as samples often contain second phases,and there are uncertainties regarding short-range order.In response to these open discussions,HEAs have been referred to recently as multiple-principle-element alloys29.It is compelling to consider similar phenomena in non-metallic systems,particularly considering existing information from entropy studies in mixed oxides.In1967Navrotsky and Kleppa showed how configurational entropy regulates the normal-to-inverse transformation in spinels,where cations transition between ordered and disordered site occupancy among the available sublattices30,31.These fundamental thermodynamic studies lead one to hypothesize that in principle,sufficient temperature would promote an additional transition to a structure containing only one sublattice with random cation occupancy.From experiment we know that before such transitions,normal materials melt,however,it is conceivable that synthetic formulations exist,which exhibit them.Inspired by research activities in the metal alloy communities and fundamental principles of thermodynamics we extend the entropy concept tofive-component oxides.With unambiguous experiments we demonstrate the existence of a new class of mixed oxides that not only contains high configurational entropy but also is indeed truly entropy stabilized.In addition,we present a hypothesis suggesting that entropy stabilization is particularly effective in a compound with ionic character.ResultsChoosing an appropriate experimental candidate.The candi-date system is an equimolar mixture of MgO,CoO,NiO,CuO and ZnO,(which we label as‘E1’)so chosen to provide the appropriate diversity in structures,coordination and cationic radii to test directly the entropic ansatz.The rationale for selection is as fol-lows:the ensemble of binary oxides should not exhibit uniform crystal structure,electronegativity or cation coordination,and there should exist pairs,for example,MgO–ZnO and CuO–NiO, that do not exhibit extensive solubility.Furthermore,the entire collection should be isovalent such that relative cation ratios can be varied continuously with electroneutrality preserved at the net cation to anion ration of unity.Tabulated reference data for each component,including structure and ionic radius,can be found in Supplementary Table1.Testing reversibility.In thefirst experiment,ceramic pellets of E1are equilibrated in an air furnace and quenched to room temperature.The temperature spanned a range from700to 1,100°C,in50-°C increments.X-ray diffraction patterns showing the phase evolution are depicted in Fig.1.After700°C,two prominent phases are observed,rocksalt and tenorite.The tenorite phase fraction reduces with increasing equilibration temperature.Full conversion to single-phase rocksalt occurs between850and900°C,after which there are no additional peaks,the background is low andflat,and peak widths are narrow in two-theta(2y)space.Reversibility is a requirement of entropy-driven transitions. Consequently,low-temperature equilibration should transform homogeneous1,000°C-equilibrated E1back to its multiphase state(and vice versa on heating).Figure1also shows a sequence of X-ray diffraction patterns for such a thermal excursion;initial equilibration at1,000°C,a second anneal at750°C,andfinally a return to1,000°C.The transformation from single phase,to multiphase,to single phase is evident by the X-ray patterns and demonstrates an enantiotropic(that is,reversible with tempera-ture32)phase transition.Testing entropy though composition variation.A composition experiment is conducted to further characterize this phase tran-sition to the random solid solution state.If the driving force is entropy,altering the relative cation ratios will influence the transition temperature.Any deviation from equimolarity will reduce the number of possible configurations O(S c¼k B log(O)), thus increasing the transition temperature.Because S c(x i)is logarithmically linked to mole fraction via B x i log(x i),the com-positional dependence is substantial.This dependency underpins our gedankenexperiment where the role of entropy can be tested by measuring the dependency of transition temperature as a function of the total number of components present,and of the composition of a single component about the equimolar formulation.The calculated entropy trends for an ideal mixture are illustrated in Fig.2b,which plots configurational entropy for a set of mixtures having N species where the composition of an individual species is changed and the others(NÀ1)are keptequimolar.Two dependencies become apparent:the entropy increases as new species are added and the maximum entropy is achieved when all the species have the same fraction.Both dependencies assume ideal random mixing.Two series of composition-varying experiments investigate the existence of these trends in formulation E1.The first experiment monitors phase evolution in five compounds,each related to the parent E1by the extraction of a single component.The sets are equilibrated at 875°C (the threshold temperature for complete solubility)for 12h.The diffraction patterns in Fig.2a show that removing any component oxide results in material with multiple phases.A four-species set equilibrated under these conditions never yields a single-phase material.The second experiment uses five individual phase diagrams to explore the configurational entropy versus composition trend.In each,the composition of a single component is varied by ±2,±6and ±10%increments about the equimolar composition while the others are kept even.Since any departure from equimolarity reduces the configurational entropy,it should increase transition temperatures to single phase,if thattransitionI n t e n s i t y2030405060702 (°)801.81,100N =5No ZnONo MgON =4No CuON =3No NiONo CoON =21,0501,000950T e m p e r a t u r e (°C )T e m p e r a t u r e (°C )T e m p e r a t u r e (°C )T e m p e r a t u r e (°C )T e m p e r a t u r e (°C )S /k B9008501,1001,0501,0009509008501,1001,0501,0009509008501,1001,0501,0009509008501,1001,0501,0009509008500.0X NX NiOX CuOX ZnOX MgoX CoO0.5 1.00.10.20.30.10.20.30.10.20.30.10.20.30.10.20.31.62223112202001111.41.21.00.80.60.40.20.0J14**********Figure 2|Compositional analysis.(a )X-ray diffraction analysis for a composition series where individual components are removed from the parent composition E1and heat treated to the conditions that would otherwise produce full solid solution.Asterisks identify peaks from rocksalt while carrots identify peaks from other crystal structures.(b )Calculated configurational entropy in an N -component solid solutions as a function of mol%of the N th component,and (c –g )partial phase diagrams showing the transition temperature to single phase as a function of composition (solvus )in the vicinity of the equimolar composition where maximum configurational entropy is expected.Error bars account for uncertainty between temperature intervals.Each phase diagram varies systematically the concentration of one element.L o g i n t e n s i t y750 °C750 °C800 °C850 °C900 °C1,000 °C2001111,000 °C 2 (°)T (200)T (002)T (110)T (200)T (002)T (110)Figure 1|X-ray diffraction patterns for entropy-stabilized oxide formulation E1.E1consists of an equimolar mixture of MgO,NiO,ZnO,CuO and CoO.The patterns were collected from a single pellet.The pellet was equilibrated for 2h at each temperature in air,then air quenched to room temperature by direct extraction from the furnace.X-ray intensity is plotted on a logarthimic scale and arrows indicate peaks associated with non-rocksalt phases,peaks indexed with (T)and with (RS)correspond to tenorite and rocksalt phases,respectively.The two X-ray patterns for 1,000°C annealed samples are offset in 2y for clarity.is in fact entropy driven.The specific formulations used are given in Supplementary Table 2.Figure 2c–g are phase diagrams of composition versus transformation temperature for the five sample sets that varied mole fraction of a single component.The diagrams were produced by equilibrating and quenching individual samples in 25°C intervals between 825and 1,125°C to obtain the T trans -composition solvus .In all cases equimolarity always leads to the lowest transformation temperatures.This is in agreement with entropic promotion,and consistent with the ideal model shown in Fig.2b.One set of raw X-ray patterns used to identify T trans for 10%MgO is given as an example in Supplementary Fig.1.Testing endothermicity .Reversibility and compositionally dependent solvus lines indicate an entropy-driven process.As such,the excursion from polyphase to single phase should be endothermic.An entropy-driven solid–solid transformation is similar to melting,thus requires heat from an external source 33.To test this possibility,the phase transformation in formulation E1can be co-analysed with differential scanning calorimetry and in situ temperature-dependent X-ray diffraction using identical heating rates.The data for both measurements are shown in Fig.3.Figure 3a is a map of diffracted intensity versus diffraction angle (abscissa)as a function of temperature.It covers B 4°of 2y space centred about the 111reflection for E1.At a temperature interval between 825and 875°C,there is a distinct transition to single-phase rocksalt structure—all diffraction events in that range collapse into an intense o 1114rocksalt peak.Figure 3b contains the companion calorimetric result where one finds a pronounced endotherm in the identical temperature window.The endothermic response only occurs when the system adds heat to the sample,uniquely consistent with an entropy-driven transformation 33.We note the small mass loss (B 1.5%)at the endothermic transition.This mass loss results from the conversion of some spinel (an intermediate phase seen by X-ray diffraction)to rocksalt,which requires reduction of 3þto 2þcations and release of oxygen to maintain stoichiometry.To address concerns regarding CuO reduction,Supplementary Fig.2shows a differential scanning calorimetry and thermal gravimetric analysis curve for pure CuO collected under the same conditions.There is no oxygen loss in the vicinity of 875°C.Testing homogeneity .All experimental results shown so far support the entropic stabilization hypothesis.However,all assume that homogeneous cation mixing occurs above the tran-sition temperature.It is conceivable that local composition fluc-tuations produce coherent clustering or phase separation events that are difficult to discern by diffraction using a laboratory sealed tube diffractometer.The solvus lines of Fig.2c–g support random mixing,as the most stable composition is equimolar (a condition only expected for ideal/regular solutions),but it is appropriate to ensure self-consistency with direct measurements.To characterize the cation distributions,extended X-ray absorption fine structure (EXAFS)and scanning transmission electron microscopy with energy dispersive X-ray spectroscopy (STEM EDS)is used to analyse structure and chemistry on the local scale.EXAFS data were collected for Zn,Ni,Cu and Co at the Advanced Photon Source 12-BM-B 34,35.The fitted data are shown in Fig.4,the raw data are given in Supplementary Fig.3.The fitted data for each element provide two conclusions:the cation-to-anion first-near-neighbour distances are identical (within experimental error of ±0.01Å)and the local structures for each element to approximately seven near-neighbour distances are similar.Both observations are only consistent with a random cation distribution.As a corroborating measure of local homogeneity,chemical analysis was conducted using a probe-corrected FEI Titan STEM with EDS detection.Thin film samples of E1,prepared by pulsed laser deposition,are the most suitable samples to make the assessment.Details of preparation are given in the methods,and X-ray and electron diffraction analysis for the film are provided in Supplementary Figs 4and 5.The sample was thinned by mechanical polishing and ion milling.Figure 5shows a collection of images including Fig.5a,the high-angle annular dark-field signal (HAADF).In Fig.5b–f,the EDS signals for the K a emission energies of Mg,Co,Ni,Cu and Zn are shown (additional lower magnification images are included in Supplementary Fig.6).All magnifications reveal chemically and structurally homogeneous material.1,100R 111R 111Mass change (%)510151,000900800700600500400300200DSC –30–20Endo DSC (mW) Exo35.536.537.52θ (°)–10010Mass100T e m p e r a t u r e (°C )T e m p e r a t u r e (°C )Figure 3|Demonstrating endothermicity.(a )In situ X-ray diffraction intensity map as a function of 2y and temperature;and (b )differential scanning calorimetry trace for formulation ‘E1’.Note that the conversion to single phase is accompanied by an endotherm.Both experiments were conducted at a heating rate of 5°C min À1.04k (Å–1)(k )×k 2 (Å–2)2ZnNiCuCo681012Figure 4|Extended X-ray absorption fine structure.EXAFS measured at Advanced Photon Source beamlime 12-BM after energy normalization and fitting.Note that the oscillations for each element occur with similar relative intensity and at similar reciprocal spacing.This suggests a similar local structural and chemical environment for each.X-ray diffraction,EXAFS and STEM–EDS probes are sensitive to 10s of nm,10s of Åand 1Ålength scales,respectively.While any single technique could be misinterpreted to conclude homogenous mixing,the combination of X-ray diffraction,EXAFS and STEM–EDS provide very strong evidence.We note,in particular,the similarity in EXAFS oscillations (both in amplitude and position)out to 12inverse angstroms.This similarly would be lost if local ordering or clustering were present.Consequently,we conclude with certainty that the cations are uniformly dispersed.DiscussionThe set of experimental outcomes show that the transition from multiple-phase to single phase in E1is driven by configurational entropy.To complete our thermodynamic understanding of this system,it is important to understand and appreciate the enthalpic penalties that establish the transition temperature.In so doing,the data set can be tested for self-consistency,and the present data are brought into the context of prior research on oxide solubility.First,we consider an equation relating the initial and final states of the proposed phase transition:MgO ðRS ÞþNiO ðRS ÞþCoO ðRS ÞþCuO ðT ÞþZnO ðW Þ¼Mg ;Ni ;Co ;Cu ;Zn ðÞO ðRS ÞFor MgO,NiO and CoO,the crystal structures of the initial and final states are identical.If we assume that solution of each into the E1rocksalt phase is ideal,the enthalpy for mixing is zero.For CuO and ZnO,there must be a structural transition to rocksalt on dissolution from tenorite and wurtzite,respectively.If we again assume (for simplicity)that the solution is ideal,the mixing energy is zero,but there is an enthalpic penalty associated with the structure transition.From Davies et al.and Bularzik et al.,we know the reference chemical potential changes for the wurtzite-to-rocksalt and the tenorite-to-rocksalt transitions of ZnO and CuO;they are 25and 22kJ mol À1,respectively 36,37.If we make the assumption that the transition enthalpies of ZnO(wurtzite)to ZnO(rocksalt E1)and CuO(tenorite)to CuO(rocksalt E1)are comparable,then the enthalpic penalty for solution into E1can be estimated.For ZnO and CuO,the transition to solid solution in a rocksalt structure involves an enthalpy change of (0.2)Á(25kJ mol À1)þ(0.2)Á(22kJ mol À1),a total of þ10kJ mol À1.This calculation is based on the productof the mol fraction of each multiplied by the reference transition enthalpy.This assumption is consistent with the report of Davies et al.who showed that the chemical potential of a particular cation in a particular structure is associated with the molar volume of that structure 36.Since the rocksalt phases of ZnO and CuO have molar volumes comparable to E1,their reference transition enthalpy values are considered suitable proxies.In comparison,the maximum theoretically expected config-urational entropy difference at 875°C (the temperature were we observe the transition experimentally)between the single species and the random five-species solid solution is B 15kJ mol À1,5kJ mol À1larger than the calculated enthalpy of transition.It is possible that the origins of this difference are related to mixing energy as the reference energy values for structural transitions to rocksalt do not capture that aspect.While the present phase diagrams that monitor T trans as a function of composition demonstrate rather symmetric behaviour about the temperature minima,it is unlikely that mixing enthalpies are zero for all constituents.Indeed,literature reports show that enthalpies of mixing between the constituent oxides in E1are finite and of mixed sign,and their magnitudes are on the same order as the 5kJ mol À1difference between our calculated predictions 36.This energy difference may be accounted for by finite and positive mixing enthalpies.Following this argument,we can achieve a self-consistent appreciation for the entropic driving force and the enthalpic penalties for solution formation in E1by considering enthalpies of the associated structural transitions and expected entropy values for ideal cation mixing.As a final test,these predictions can be compared with experiment,specifically by calculating the magnitude of the endotherm observed by DSC at the transition from multiple-phase to single-phase states.Doing so we find a value B 12kJ mol À1(with an uncertainty of ±2kJ mol À1).While we acknowledge the challenge of quantitative calorimetry,we note that this experimental result is intermediate to and in close agreement with the predicted values.Compared with metallic alloys,the pronounced impact of entropy in oxides may be surprising given that on a per-atom basis the total disorder per volume of an oxide seems be lower than in a high-entropy alloy,as the anion sublattice is ordered (apart from point defects).The chemically uniform sublattice is perhaps the key factor that retains cation configurational entropy.As an illustration,consider a comparison between random metal alloys and random metal oxide alloys.Begin by reviewing the case of a two-component metallic mixture A–B.If the mixture is ideal,the energy of interaction E A–B ¼(E A–A þE B–B )/2,there is no enthalpic preference for bonding,and entropy regulates solution formation.In this scenario,all lattice sites are equivalent and configurational entropy is maximized.This situation,however,never occurs as no two elements have identical electronegativity and radii values.Figure 6a illustrates a two-component alloy scenario A–B where species B is more electronegative than A.Consequently,the interaction energies E A–A ,E B–B and E A–B will be different.A random mixture of A–B will produce lattice sites with a distribution of first near neighbours,that is,species A coordinated to 4-B atoms,2-A and 2-B atoms,etc y Different coordinations will have different energy values and the sites are no longer indistinguishable.Reducing the number of equivalent sites reduces the number of possible configurations and S .Now consider the same two metallic ions co-populating a cation sublattice,as in Fig.6b.In this case,there is always an intermediate anion separating neighbouring cation lattice sites.Again,in the limiting case where only first near neighboursareFigure 5|STEM–EDS analysis of E1.(a )HAADF image.Panels labelled as Zn,Ni,Cu,Mg and Co are intensity maps for the respective characteristic X-rays.The individual EDS maps show uniform spatial distributions for each element and are atomically resolved.considered,every cation lattice site is ‘identical’because each has the same immediate surroundings:the interior of an oxygen octahedron.Differentiation between sites is only apparent when the second near neighbours are considered.From the configura-tional disorder perspective,if each cation lattice site is identical,and thus energetically similar to all others,the number of microstates possible within the macrostate will approach the maximum value.This crystallographic argument is based on the limiting case where first-near-neighbour interactions predominate the energy landscape,which is an imperfect approximation.Second and third near neighbours will influence the distribution of lattice site energies and the number of equivalent microstates—but the impact will be the same in both scenarios.A larger number of equivalent sites in a crystal with an intermediate sublattice will increase S and expand the elemental diversity containable in a single solid solution and to lower the temperature at which the transition to entropic stabilization occurs.We acknowledge the hypothesis nature of this model at this time,and the need for a rigorous theoretical exploration.It is presented currently as a possibility and suggestion for future consideration and testing.We demonstrate that configurational disorder can promote reversible transformations between a poly-phase mixture and a homogeneous solid solution of five binary oxides,which do not form solid solutions when any of the constituents are removed provided the same thermal budget.The outcome is representative of a new class of materials called ‘entropy-stabilized oxides’.While entropic effects are known for oxide systems,for example,random cation occupancy in spinels 30,order–disorder transfor-mations in feldspar 38,and oxygen nonstoichiometry in layered perovskites 39,the capacity to actively engineer configurational entropy by composition,to stabilize a quinternary oxide with a single cation sublattice,and to stabilize unusual cation coordination values is new.Furthermore,these systems provide a unique opportunity to explore the thermodynamics and structure–property relationships in systems with extreme configurational disorder.Experimental efforts exploring this composition space are important considering that such compounds will be challenging to characterize with computational approaches minimizing formation energy (for example,genetic algorithms)or with adhoc thermodynamic models (for example,CALPHAD,cluster expansion)6.We expect entropic stabilization in systems where near-neighbour cations are interrupted by a common intermediateanion (or vice versa),which includes broad classes of chalcogenides,nitrides and halides;particularly when covalent character is modest.The entropic driving force—engineered by cation composition—provides a departure from traditional crystal-chemical principles that elegantly predict structural trends in the major ternary and quaternary systems.A companion set of structure–property relationships that predict new entropy-stabilized structures with novel cation incorporation await discovery and exploitation.MethodsSolid-state synthesis of bulk materials .MgO (Alfa Aesar,99.99%),NiO (Sigma Aldrich,99%),CuO (Alfa Aesar,99.9%),CoO (Alfa Aesar,99%)and ZnO (Alfa Aesar 99.9%)are massed and combined using a shaker mill and 3-mm diameter yttrium-stabilized zirconia milling media.To ensure adequate mixing,all batches are milled for at least 2h.Mixed powders are then separated into 0.500-g samples and pressed into 1.27-cm diameter pellets using a uniaxial hydraulic press at 31,000N.The pellets are fired in air using a Protherm PC442tube furnace.Temperature evolution of phases .Ceramic pellets of E1are equilibrated in an air furnace and quenched to room temperature by direct extraction from the hot zone.Phase analysis is monitored by X-ray diffraction using a PANalytical Empyrean X-ray diffractometer with Bragg-Brentano optics including programmable diver-gence and receiving slits to ensure constant illumination area,a Ni filter,and a 1-D 128element strip detector.The equivalent counting time for a conventional point detector would be 30s per point at 0.01°2y increments.Note that all X-ray are collected using substantial counting times and are plotted on a logarithmic scale.To the extent knowable using a laboratory diffractometer,the high-temperature samples are homogeneous and single phase:there are no additional minor peaks,the background is low and flat,and peak widths are sharp in two-theta (2y )space.Temperature-dependent diffraction data are collected with PANalytical Empyrean X-ray diffractometer with Bragg-Brentano optics includingprogrammable divergence and receiving slits to ensure constant illumination area,a Ni filter,and a 1-D 256element strip detector.The samples are placed in a resistively heated HTK-1200N hot stage in air.The samples are ramped at a constant rate of 5°C min À1with a theta–two theta pattern captured every 1.5min.Calorimetry data are collected using a Netzsch STA 449F1Jupiter system in a Pt crucible at 5°C min À1in flowing air.Determining solvus lines .Five series of powders are mixed where the amount of one constituent oxide is varied from the parent mixture E1.Supplementary Table 2lists the full set of samples synthesized for this experiment.Each individual sample is cycled through a heat-soak-quench sequence at 25°C increments from 850°C up to 1,150°C.The soak time for each cycle is 2h,and samples are then quenched to room temperature in o 1min.After the quenching step for each cycle,samples are immediately analysed for phase identification using a PANalytical Empyrean X-ray diffractometer using the conditions identified above.If more than one phase is present,the sample would be put through the next temperature cycle.The temperature at which the structure is determined to be pure rocksalt,with no discernable evidence of peak splitting or secondary phases,is deemed the transition temperature as a function of composition.Supplementary Fig.1shows an example of the collected X-raypatterns after each cycle using the E1L series with þ10%MgO.Once single phase is achieved,the sample is removed from the sequence.Note that this entire experiment is conducted two times.Initially in 50°C increments and longer anneals,and to ensure accuracy of temperature values and reproducibility,a second time using shorter increments and 25°C anneals.Findings in both sets are identical to within experimental error bar values.In the latter case,error bars correspond to the annealing interval value of 25°C.In the main text relating to Fig.2a we note that in addition to small peaks from second phases,X-ray spectra for N ¼4samples with either NiO or MgO removed show anisotropic peak broadening in 2y and skewed relative intensities where I (200)/I (111)is less than unity.This ratio is not possible for the rocksalt structure.Supplementary Table 3shows the result of calculations of structure factors for a random equimolar rocksalt oxide with composition E1.Calculations show that the 200reflection is the strongest,and that the experimentally measured relative intensities of 111/200are consistent with calculations.We use this information as a means too best assess when the transition to single phase occurs since the most likely reason for the skewed relative intensity is an incomplete conversion to the single-phase state.This dependency is highlighted in Supplementary Fig.1.X-ray absorption fine structure .X-ray absorption fine structure (XAFS)is made possible through the general user programme at the Advanced Photon Source in Lemont,IL (GUP-38672).This technique provides a unique way to probe the local environment of a specific element based on the interference between an emitted core electron and the backscattering from surrounding species.XAFS makes no assumption of structure symmetry or elemental periodicity,making it an ideal means to study disordered materials.During the absorption process,coreelectronsBFigure 6|Binary metallic compared with a ternary oxide.A schematic representation of two lattices illustrating how the first-near-neighbour environments between species having different electronegativity (the darker the more negative charge localized)for (a )a random binary metal alloy and (b )a random pseudo-binary mixed oxide.In the latter,near-neighbour cations are interrupted by intermediate common anions.。


星形胶质细胞rna提取 ## English Response: ##。
Materials.Brain tissue.Sterile dissecting instruments.RNAlater (Sigma-Aldrich)。
QIAzol Lysis Reagent (Qiagen)。
Chloroform.Isopropanol (cold)。
75% Ethanol (cold)。
RNase-Free Water.Microcentrifuge and tubes.Procedure.1. Harvest the brain tissue. Euthanize the animal and remove the brain. Carefully dissect the brain tissue of interest (e.g., hippocampus, cortex).2. Immerse the tissue in RNAlater. Transfer the dissected tissue to a tube containing RNAlater. Incubate overnight at 4°C.3. Lyse the tissue. Centrifuge the RNAlater-treated tissue at 12,000 x g for 10 minutes at 4°C. Remove the supernatant and resuspend the pellet in QIAzol Lysis Reagent. Homogenize the tissue using a tissue homogenizer or sonication.4. Extract the RNA. Add chloroform to the homogenized tissue and mix vigorously. Centrifuge at 12,000 x g for 15 minutes at 4°C. Transfer the upper aqueous phase to a newtube.5. Precipitate the RNA. Add isopropanol to the aqueous phase and mix. Centrifuge at 12,000 x g for 10 minutes at 4°C. Wash the RNA pellet with cold 75% ethanol.6. Resuspend the RNA. Centrifuge the RNA pellet at12,000 x g for 5 minutes at 4°C. Remove the ethanol andair-dry the pellet. Resuspend the RNA in RNase-Free Water.Quantification and Quality Assessment.Quantify the RNA concentration using a spectrophotometer (e.g., Nanodrop).Assess the RNA quality using an Agilent Bioanalyzer or similar platform.## 中文回答,##。

邹怡茜,陈海强,潘卓官,等. 超高压耦合热处理过程对鳙鱼鱼糜凝胶特性及水分迁移的影响[J]. 食品工业科技,2023,44(23):70−79. doi: 10.13386/j.issn1002-0306.2023020083ZOU Yiqian, CHEN Haiqiang, PAN Zhuoguan, et al. Effects of Gel Properties and Water Migration during Ultra-High Pressure Coupled Heat Treatment on Bighead Carp Surimi[J]. Science and Technology of Food Industry, 2023, 44(23): 70−79. (in Chinese with English abstract). doi: 10.13386/j.issn1002-0306.2023020083· 研究与探讨 ·超高压耦合热处理过程对鳙鱼鱼糜凝胶特性及水分迁移的影响邹怡茜1,陈海强1,2,潘卓官1,周爱梅1,*(1.华南农业大学食品学院,广东广州 510642;2.阳江职业技术学院,广东阳江 529500)摘 要:为了阐明超高压耦合热处理过程中鳙鱼鱼糜凝胶特性变化的机制,本文探究了超高压耦合热处理(300 MPa/5 min ,40 ℃/30 min ,90 ℃/20 min )过程中鳙鱼鱼糜凝胶特性、蛋白质结构及水分迁移的变化,并进行聚类热图和Pearson 相关性分析。
结果表明,超高压耦合热处理能显著改善鳙鱼鱼糜的凝胶特性(P <0.05)。
随着超高压,超高压结合一段热处理,超高压结合二段热处理过程的进行,鳙鱼鱼糜凝胶的凝胶强度、质构、白度呈上升趋势,其中,较常压处理样品(0.1P ),经超高压耦合热处理(300PSH )的鱼糜凝胶强度和白度分别增加了477.75%、43.38%。

Cat. #R045AProduct ManualPrimeSTAR® Max DNAPolymeraseFor Research Usev201510DaTable of ContentsI. Description (3)II. Components (3)III. Storage (3)IV. General Composition of PCR Reaction Mixture (3)V. PCR Conditions (4)VI. Optimization of Parameters (6)VII. Features (7)VIII. Electrophoresis, Cloning, and Sequencing of Amplified Products (12)IX. Troubleshooting (12)X. Related Products (13)I. DescriptionPrimeSTAR Max DNA Polymerase is a unique high-performance DNA polymerase that possesses the fastest extension speed available, along with the extremely high accuracy, high sensitivity, high specificity, and high fidelity of PrimeSTAR HS DNA Polymerase. High priming efficiency and extension efficiency greatly reduces the time required for annealing and extension steps, facilitating exceptionally fast high-speed PCR reactions. In addition, standardization of extension step time makes PrimeSTAR Max DNA Polymerase suitable for reactions with large amounts of template DNA that would ordinarily be difficult to amplify. Furthermore, an antibody-mediated hot start formulation prevents false initiation events during the reaction assembly due to mispriming and primer digestion. Since PrimeSTAR Max DNA Polymerase is configured as a 2-fold premix containing reaction buffer and dNTP mixture, it allows quick preparation of reactions and is useful for high-throughput applications.II. Components (for 100 reactions, 50 μl volume)PrimeSTAR Max Premix (2X) 625 μl x 4* Containing 2 mM Mg2+ and 0.4 mM each dNTPIII. Storage–20℃Note: Repeated freeze-thaw of the Premix may reduce its activity.IV. General Composition of PCR Reaction MixtureFinal conc.PrimeSTAR Max Premix (2X)25 μl1XPrimer 110 - 15 pmol0.2 - 0.3 μMPrimer 210 - 15 pmol0.2 - 0.3 μMTemplate< 200 ng*Sterile distilled water to reaction volume of 50 μl*: Refer to VI. Optimization of ParametersCaution:The PCR reaction mixture can be prepared at room temperature. However,keep each of the reaction components on ice during the preparation process.V. PCR ConditionsWhen performing rapid amplification protocols using PrimeSTAR Max DNAPolymerase, 3-step reactions are recommended for best results and longestamplification products.(A) For reactions in which the quantity of template is 200 ng / 50 μl or less:*98℃10 sec.55℃ 5 sec. or 15 sec.30 - 35 cycles72℃ 5 sec./kb(B) For reactions in which the quantity of template exceeds 200 ng / 50 μl:*98℃10 sec.55℃ 5 sec. or 15 sec.30 - 35 cycles [3-step PCR]72℃30 - 60 sec./kbor98℃10 sec.30 - 35 cycles [2-step PCR]68℃30 - 60 sec./kb*: For rapid amplification protocols (extension step of 5 to 10 sec./kb) withcDNA as template, use a quantity of template that is equal to or less thanthe equivalent of 125 ng of total RNA / 50 μl reaction.If larger quantites of cDNA template are desired, by setting a longerextension time (up to 1 min./kb), it is possible to use up to the equivalentof 750 ng total RNA / 50 μl reaction.See VII.C. Template Quantity and Reaction Speed Using cDNA as Template.・ Denaturing conditions: An initial denaturation step is not necessary for some PCRenzymes, including the PrimeSTAR polymerase series;98℃ for 10 seconds is sufficient for complete denaturation.During cycling, denaturation at 98℃ for 5 to 10 sec. isrecommended.Denaturation at 94℃ is also possible, but the time shouldbe extended to 10 to 15 sec.・ Annealing temperature: Use 55℃ as the default annealing temperature.・ Annealing time: For primers that are 25-mer or shorter:For primer T m values (calculated by the formula below) of55℃ or greater, anneal for 5 sec.For primer T m values (calculated by the formula below)less than 55℃, anneal for 15 sec.For primers longer than 25-mers:Use an annealing time of 5 sec.*Tm value calculation:Tm (℃) = 2(NA + NT) + 4(NC + NG) - 5where N represents the number of primer nucleotides having thespecified identity (A, T, C, or G)・ Final elongation: This step is typically recommended for Taq polymerase, but isnot always necessary with PrimeSTAR Max polymerase.Important note:Because the priming efficiency of PrimeSTAR Max DNA Polymerase is extremely high, use an annealing time of 5 sec. or 15 sec. Longer annealing times may cause smearing of PCR products visible during electrophoresis analysis.If smearing occurs when performing a 3-step PCR protocol, try a 2-step PCR protocol. See VI. Optimization of Parameters and IX. Troubleshooting.VI. Optimization of ParametersIn order to obtain the best PCR results, it is important to optimize the PrimeSTAR Max DNAPolymerase reaction parameters to fully utilize the enzyme's properties and advantages.(1) Template DNARecommended quantities of template DNA (50 μl reaction) for rapid amplificationprotocols (extension step of 5 sec./kb):Human genomic DNA 5 ng - 200 ngE. coli genomic DNA 100 pg - 200 ngλDNA 10 pg - 10 ngPlasmid DNA 10 pg - 1 ngWhen using more than 200 ng of DNA as template in a 50 μl reaction, use anextension time of 30 to 60 sec./kb for best results.For rapid amplification protocols (extension time of 5 to 10 sec./kb) with cDNA astemplate, set the template cDNA quantity to ≦the equivalent of 25 to 125 ng total RNAper 50 μl reaction.See VI. C. Template Quantity and Reaction Speed Using cDNA as Template.Do not use templates containing uracil, such as bisulfite-treated DNA.(2) Amplified Product SizesAmplification product sizes using an extension time of 5 sec./kb (for genomic DNAtemplates) or 5 to 10 sec./kb (for cDNA templates):Human genomic DNA up to 6 kbE. coli genomic DNA up to 10 kbcDNA up to 6 kbλDNA up to 15 kbWhen amplifying targets in excess of these lengths, try using an extension time of 15to 30 sec./kb. In such instances, amplification is affected by the quantity, quality, andsequence composition of the template.(3) Primer and PCR ConditionsSelect primer sequences using primer design software such as OLIGO Primer AnalysisSoftware (Molecular Biology Insights, Inc.).For general amplification, 20 to 25-mer primers are suitable. When amplifying longerproducts, the use of 25 to 30-mer primers may improve results. See section V. PCRConditions.Do not use inosine-containing primers with PrimeSTAR Max DNA Polymerase.(4) Annealing conditionsSelect annealing conditions as described in V. PCR Conditions. If low product yieldoccurs, try the following:<If smearing and/or extra bands appear on agarose electrophoresis gels>(1) Shorten the annealing time. If performing at 15 sec., set to 5 sec.(2) If the annealing step has already been set to 5 sec., raise the annealingtemperature to 58℃ - 63℃.(3) Perform 2-step PCR.<If the target product is not amplified>(1) Lengthen the annealing time. If performing at 5 sec., set to 15 sec.(2) Lower the annealing temperature to 50℃ - 53℃.Good amplification is observed for products up to 6 kb in length using an extension time of 10 sec. and for products up to 8 kb in length using an extension time of 30 sec.When λDNA is used as template, extension time of 5 sec./kb may be suitable.(2) With human genomic DNA as template, amplification of products ranging in sizefrom 0.5 kb to 7.5 kb was performed using an annealing time of 5 sec. and an extension time of either 10 or 30 sec.Template Human genomic DNA [100 ng / 50 μl reaction]Thermal cyclerTaKaRa PCR Thermal Cycler Dice(Not available in all geographic locations. Check for availability in your region.)PCR conditions98℃10 sec.55℃ 5 sec.30 cycles72℃10 or 30 sec.M: λ-Hin d III digest[Extension time: 30 sec.]M1246810M (kb)8M1246(kb)10M [Extension time: 10 sec.]VII. FeaturesA. Rapid Amplification(1) With λDNA as template, amplification of products ranging in size from 1 to 10 kbwas performed using an annealing time of 5 sec. and an extension time of either 10 or 30 sec.Template λDNA [1 ng/50 μl reaction]Thermal cyclerTaKaRa PCR Thermal Cycler Dice™(Not available in all geographic locations. Check for availability in your region.)PCR conditions98℃10 sec.55℃ 5 sec.30 cycles72℃10 or 30 sec.M: λ-Hin d III digest[Extension time: 10 sec.]M 0.5123467.5M(kb)[Extension time: 30 sec.]M 0.5123467.5M (kb)Good amplification is observed for products up to 4 kb in length using an extension time of 10 sec, and for products up to 6 kb in length using an extension time of 30 sec. With human genomic DNA as template, an extension time setting of 5 sec./kb may be suitable.(3) With cDNA template, amplification of products ranging in size from 1 kb to 6 kb wasperformed using an annealing time of 15 sec. and an extension time of either 10 or 30 sec.Template cDNA [equivalent to 100 ng total RNA) / 50 μl reaction]Thermal cyclerTaKaRa PCR Thermal Cycler Dice(Not available in all geographic locations. Check for availability in your region.)PCR conditions98℃10 sec.55℃15 sec.30 cycles72℃10 or 30 sec.M: λ-Hind III digest[Extension time]M 1246M 1246M (kb)10 sec.30 sec.Good amplification was observed for products up to 2 kb in length using anextension time of 10 sec. and for products up to 4 kb using an extension time of 30 sec. With cDNA template, an extension time of 5 to 10 sec./kb is required.B. Length of amplification productsWith λDNA, E. coli genomic DNA, human genomic DNA, or cDNA as the template, amplification sizes of various DNA fragments were examined using an annealing time of 5 sec. or 15 sec. and an extension time of 5 sec./kb (genomic DNA) or 10 sec./kb (cDNA).Template:λDNA 1 ng E. coli genomic DNA 50 ng Human genomic DNA 100 ng cDNA equivalent to 100 ng total RNAThermal cycler:TaKaRa PCR Thermal Cycler Dice PCR conditions: 98℃10 sec.55℃ 5 or 15 sec.30 cycles 72℃ 5 (or 10) sec./kbGood amplification of products up to 6 kb in length was observed using an extension time of 5 sec./kb.[ Human genomic DNA ]M 246M1(kb)0.57.53M: λ-Hin d III digestGood amplification of products up to 6 kb in length was observed using an extension time of 10 sec./kb.[ cDNA ][λDNA ]Good amplification of products up to 15 kb inlength was observed using an extension time of 5 sec./kb.M: λ-Hin d III digestM2468101215M 1(kb)Good amplification of products up to 10 kb in length was observed using an extension time of 5 sec./kb.[ E. coli genomic DNA ]M246810M: λ-Hind III digestM (kb)M2468M 1(kb)M: λ-Hind III digestC. Template quantity and reaction rate using cDNA as templateAmplification of transferrin receptor (TFR) 4 kb in length was performed with cDNA as template. cDNA was obtained by reverse transcription of various amounts of total RNA, as indicated. The extension times were set to 20 sec (5 sec./kb), 2 min (30 sec./kb) or 4 min (1 min./kb), and the amplification efficiencies were compared.Template quantity (50 μl reaction)1 : cDNA equivalent to 25 ng total RNA 2 : cDNA equivalent to 50 ng total RNA 3 : cDNA equivalent to 125 ng total RNA 4 : cDNA equivalent to 250 ng total RNAM 1234567M 1234567M 1234567M5 : cDNA equivalent to 500 ng total RNA6 : cDNA equivalent to 750 ng total RNA7 : cDNA equivalent to 1 μg total RNA M : λ-Hind III digest1 min./kb5 sec./kb30 sec./kbM : λ-Hind III digestM 1234M 1234M 1234M 1234MHumangenomic DNA E. coli genomic DNAλDNAPlasmidTemplate quantity*:Lane 1 Lane 2 Lane 3 Lane 4Human genomic DNA 100 pg 1 ng 10 ng 100 ng E. coli genomic DNA 1 pg 10 pg 100 pg 1 ng λDNA100 fg 1 pg 10 pg 100 pg Plasmid DNA100 fg1 pg10 pg100 pgFor rapid amplification protocols using an extension time of 5 sec./kb, it is necessaryto use cDNA template that is ≦ the equivalent of 125 ng total RNA / 50 μl reaction. When using longer extension times (up to 1 min./kb), the quantity of cDNA template can be increased up to the equivalent of 750 ng total RNA / 50 μl reaction.D. SensitivityWith various amounts of human genomic DNA, E. coli genomic DNA, λDNA, orplasmid DNA as template, sensitivity was examined when amplification of a 4 kb DNA fragment was performed using an extension time of 20 sec.Thermal cycler TaKaRa PCR Thermal Cycler Dice PCR conditions98℃55℃72℃10 sec.5 sec.20 sec.30 cycles *: Observed limit of detection indicated by underline.E. AccuracyThe fidelity of PrimeSTAR Max DNA Polymerase was examined by analysis of sequenc-ing data.[ Method ] Eight arbitrarily selected GC-rich regions were amplified with PrimeSTARMax DNA Polymerase or other DNA polymerases, using Thermus ther-mophilus HB8 genomic DNA as template.PCR products (approx. 500 bp each) were each cloned into a suitable plas-mid. Multiple clones were selected per respective amplification productand were subjected to sequence analysis.[ Result ] Sequence analysis of DNA fragments amplified using PrimeSTAR Max DNAPolymerase demonstrated only 9 mismatched bases per 230,129 total bases. This is higher fidelity than an alternative high-fidelity enzyme from Company A, and 10-fold higher fidelity than Taq DNA polymerase.*: Out of 230,129 analyzed bases that were amplified using PrimeSTARMax DNA Polymerase, only 9 base errors occurred.Fidelity comparison of each enzymem u t a t i o n f r e q u e n c y (%)0.060%0.050%0.040%0.030%0.020%0.010%0.000%PrimeSTARHSPrimeSTAR Max *Company A High FidelityEnzymePfuTaqVIII.Electrophoresis, Cloning, and Sequencing of Amplified Products1) ElectrophoresisTAE Buffer is recommended for agarose gel electrophoresis of amplified products that areobtained using PrimeSTAR Max DNA Polymerase.Note : Use of TBE Buffer may result in DNA band patterns that are enlarged at the bottom of the gel.2) Termini of amplified productsMost PCR products amplified with PrimeSTAR Max DNA Polymerase have blunt-endtermini. Accordingly, they can be cloned directly into blunt-end vectors. If necessary,phosphorylate the amplified products before cloning. Use of Mighty Cloning Reagent Set(Blunt End) (Cat. #6027)is recommended for cloning into a blunt-end vector.3) Restriction enzyme reactionPrior to performing restriction enzyme digestion of amplified PCR products, remove alltraces of PrimeSTAR Max DNA Polymerase from the reaction mixture by phenol/chloro-form extraction or by using NucleoSpin Gel and PCR Clean-up (Cat. #740609.10/.50/.250).Particularly for 3'-protruding restriction enzymes such as Pst I, the 3'-protruding terminiproduced by these enzymes may be deleted by 3' → 5' exonuclease activity of PrimeSTARMax DNA Polymerase, if residual polymerase remains present in the restriction digest reaction.4) Direct sequencingPerform phenol/chloroform extraction of PCR products prior to direct sequencing toensure inactivation of 3' → 5' exonuclease activity. Alternatively, NucleoSpin Gel and PCRClean-up (Cat. #740609.10/.50/.250) may be used to purify DNA prior to sequencing.IX. TroubleshootingEvent Possible causes ActionNo amplification orpoor amplification efficiency Extension time Set to 10 to 60 sec./kb *Number of cycles Set to 35 to 40 cycles.Annealing time Set to 15 sec.Annealing temperature Lower by 2℃ per trialReaction volume Use 25 μl.Purity and quantity oftemplate DNAUse an appropriate amount of template DNA.Purify the template DNA*.Primer concentration Use 0.2 - 0.5 μM (final conc.).Electrophoresis analysis shows smeared band(s) or extra band(s)Annealing time Set to 5 sec.AnnealingtemperatureRaise by 2℃ per trial up to 63℃.Try 2-step PCR.Template DNAquantityUse an appropriate amount of template DNA.Do not use more than necessary.Number of cycles Set to 25 to 30 cycles.Primer concentration Use at a final concentartion of 0.2 - 0.3 μM.*: When using crude samples containing large quantities of RNA, such as samples prepared by thermal lysis, improved results may be achieved by setting the extension time to60 sec./kb.X. Related ProductsPrimeSTAR® HS DNA Polymerase(Cat. #R010A/B)PrimeSTAR® HS (Premix) (Cat. #R040A)PrimeSTAR® GXL DNA Polymerase (Cat. #R050A/B)PrimeSTAR® Mutagenesis Basal Kit (Cat. #R046A)*NucleoSpin Gel and PCR Clean-up (Cat. #740609.10/.50/.250)Mighty Cloning Reagent Set (Blunt End) (Cat. #6027)TaKaRa PCR Thermal Cycler Dice™ Gradient/Standard (Cat. #TP600/TP650)*TaKaRa PCR Thermal Cycler Dice™ Touch (Cat. #TP350)** : Not available in all geographic locations. Check for availability in your region.PrimeSTAR is a registered trademark of TAKARA BIO INC.Thermal Cycler Dice is a trademark of TAKARA BIO INC.NOTE :This product is for research use only. It is not intended for use in therapeutic or diagnostic procedures for humans or animals. Also, do not use this product as food, cosmetic, orhousehold item, etc.Takara products may not be resold or transferred, modified for resale or transfer, or usedto manufacture commercial products without written approval from TAKARA BIO INC.If you require licenses for other use, please contact us by phone at +81 77 565 6973 orfrom our website at .Your use of this product is also subject to compliance with any applicable licensingrequirements described on the product web page. It is your responsibility to review,understand and adhere to any restrictions imposed by such statements.All trademarks are the property of their respective owners. Certain trademarks may not be registered in all jurisdictions.。


收稿日期:2023-07-23基金项目:海南省自然科学基金(318QN189);海南省教育厅项目(Hnky2021-19,Qhys2021-248)作者简介:李雨欣(1999-),女,在读硕士生,研究方向为植物耐盐机理,E-mail:**********************通信作者:周扬(1988-),男,博士,副教授,研究方向为植物抗逆机理,E-mail:*********************.cn广东农业科学2023,50(10):85-96Guangdong Agricultural SciencesDOI:10.16768/j.issn.1004-874X.2023.10.010李雨欣,王鹏,刘雯,周扬. 海马齿NHX 基因家族的鉴定及表达分析[J]. 广东农业科学,2023,50(10):85-96.海马齿NHX 基因家族的鉴定及表达分析李雨欣,王 鹏,刘 雯,周 扬〔海南大学 热带农林学院(农业农村学院、乡村振兴学院)/海南省热带园艺作物品质调控重点实验室,海南 海口 570228〕摘 要:【目的】海马齿(Sesuvium portulacastrum L.)是典型的海岸植物和红树林伴生植物,能够固沙护岸,在海水中可以正常生长,具有极强的耐盐性。
Na +/H +逆向转运蛋白(Na +/H + exchange,NHX)在植物耐盐和生长发育中起关键作用,为了解NHX 在海马齿耐盐过程中的作用,对海马齿NHX 基因家族进行鉴定和分析。
【方法】采用生物信息学方法从海马齿全长转录组数据中鉴定NHXs 成员,对其蛋白理化性质、保守基序和进化关系进行分析,并利用荧光定量PCR 技术研究盐胁迫下NHXs 成员的表达模式。
【结果】从海马齿中共鉴定出12个SpNHX 基因,命名为SpNHX1~SpNHX12。
SpNHXs 基因编码的氨基酸长度为276~554 aa,分子量为31.22~61.21 kD,等电点为5.55~8.64,不稳定指数为32.14~50.54,均为疏水性蛋白。

第一章绪论一简答题1. 21世纪是生命科学的世纪。
20世纪后叶分子生物学的突破性成就,使生命科学在自然科学中的位置起了革命性的变化。
试阐述分子生物学研究领域的三大基本原则,三大支撑学科和研究的三大主要领域?答案:(1)研究领域的三大基本原则:构成生物大分子的单体是相同的;生物遗传信息表达的中心法则相同;生物大分子单体的排列(核苷酸,氨基酸)导致了生物的特异性。
(2)三大支撑学科:细胞学,遗传学和生物化学。
(3)研究的三大主要领域:主要研究生物大分子结构与功能的相互关系,其中包括DNA和蛋白质之间的相互作用;激素和受体之间的相互作用;酶和底物之间的相互作用。
2. 分子生物学的概念是什么?答案:有人把它定义得很广:从分子的形式来研究生物现象的学科。
但是这个定义使分子生物学难以和生物化学区分开来。
另一个定义要严格一些,因此更加有用:从分子水平来研究基因结构和功能。
从分子角度来解释基因的结构和活性是本书的主要内容。
3 二十一世纪生物学的新热点及领域是什么?答案:结构生物学是当前分子生物学中的一个重要前沿学科,它是在分子层次上从结构角度特别是从三维结构的角度来研究和阐明当前生物学中各个前沿领域的重要学科问题,是一个包括生物学、物理学、化学和计算数学等多学科交叉的,以结构(特别是三维结构)测定为手段,以结构与功能关系研究为内容,以阐明生物学功能机制为目的的前沿学科。
这门学科的核心内容是蛋白质及其复合物、组装体和由此形成的细胞各类组分的三维结构、运动和相互作用,以及它们与正常生物学功能和异常病理现象的关系。
分子发育生物学也是当前分子生物学中的一个重要前沿学科。
人类基因组计划,被称为“21世纪生命科学的敲门砖”。
“人类基因组计划”以及“后基因组计划”的全面展开将进入从分子水平阐明生命活动本质的辉煌时代。
目前正迅速发展的生物信息学,被称为“21世纪生命科学迅速发展的推动力”。
尤应指出,建立在生物信息基础上的生物工程制药产业,在21世纪将逐步成为最为重要的新兴产业;从单基因病和多基因病研究现状可以看出,这两种疾病的诊断和治疗在21世纪将取得不同程度的重大进展;遗传信息的进化将成为分子生物学的中心内容”的观点认为,随着人类基因组和许多模式生物基因组序列的测定,通过比较研究,人类将在基因组上读到生物进化的历史,使人类对生物进化的认识从表面深入到本质;研究发育生物学的时机已经成熟。
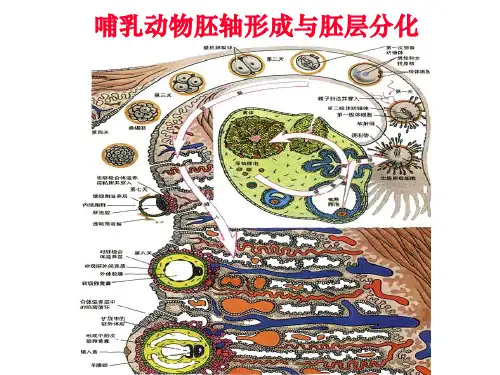

R E V I E W A R T I C L ECandida glabrata ,Candida parapsilosis and Candida tropicalis:biology ,epidemiology ,pathogenicityand antifungal resistanceS ´onia Silva 1,Melyssa Negri 1,Mariana Henriques 1,Ros ´ario Oliveira 1,David W.Williams 2&Joana Azeredo 11Institute for Biotechnology and Bioengineering,Universidade do Minho,Campus de Gualtar,Braga,Portugal;and 2Tissue Engineering &Reparative Dentistry,School of Dentistry,Heath Park,Cardiff,UKCorrespondence:Mariana Henriques,Institute for Biotechnology andBioengineering,Universidade do Minho,Campus de Gualtar 4710-057,Braga,Portugal.Tel.:1351253604408;fax:1351253604429;e-mail:mcrh@deb.uminho.pt Received 29December 2010;revised 31March 2011;accepted 6May 2011.DOI:10.1111/j.1574-6976.2011.00278.x Editor:Martin KupiecKeywordsCandida species;candidosis;epidemiology;virulence factors;antifungal resistance.AbstractThe incidence of infections caused by Candida species (candidosis)has increased considerably over the past three decades,mainly due to the rise of the AIDS epidemic,an increasingly aged population,higher numbers of immunocompro-mised patients and the more widespread use of indwelling medical devices.Candida albicans is the main cause of candidosis;however,non-C.albicans Candida (NCAC)species such as Candida glabrata ,Candida tropicalis and Candida parapsilosis are now frequently identified as human pathogens.The apparent increased emergence of these species as human pathogens can be attributed to improved identification methods and also associated with the degree of diseases of the patients,the interventions that they were subjected and the drugs used.Candida pathogenicity is facilitated by a number of virulence factors,most importantly adherence to host surfaces including medical devices,biofilm forma-tion and secretion of hydrolytic enzymes (e.g.proteases,phospholipases and haemolysins).Furthermore,despite extensive research to identify pathogenic factors in fungi,particularly in C.albicans ,relatively little is known about NCAC species.This review provides information on the current state of knowledge on the biology,identification,epidemiology,pathogenicity and antifungal resistance of C.glabrata ,C.parapsilosis and C.tropicalis .IntroductionIn last 30years there has been a significant increase in the incidence of fungal infections in humans (Lass-Fl¨o rl,2009).Such infections may either be superficial,affecting the skin,hair,nails and mucosal membranes,or systemic,involving major body organs (Ruping et al .,2008).A number of factors have been implicated in this increased occurrence of fungal disease,but it is generally accepted that the increased and widespread use of certain medical practices,such as immunosuppressive therapies,invasive surgical procedures and use of broad-spectrum antibiotics are significant (Sa-maranayake et al .,2002;Hagerty et al .,2003;Kojic &Darouiche,2004).Of the fungi regarded as human pathogens,the members of the genus Candida are the most frequently recovered from human fungal infection.The Candida genus contains over 150heterogeneous species (Calderone,2002),but only aminority have been implicated in human candidosis.Addi-tionally,it is known that approximately 65%of Candida species are unable to grow at a temperature of 371C,which precludes these species from being successful pathogens or indeed commensals of humans (Calderone,2002).Of the Candida species isolated from humans,Candida albicans is the most prevalent under both healthy and disease (Calderone,2002;Samaranayake et al .,2002)condi-tions.However,while mycological studies have shown that C.albicans represents over 80%of isolates from all forms of human candidosis (Calderone,2002)in the last two decades,the number of infections due to non-C.albicans Candida (NCAC)species has increased significantly (Kauffman et al .,2000;Manzano-Gayosso et al .,2008;Ruan &Hsueh,2009).The apparent increased involvement of NCAC species in human candidosis may partly be related to improvements in diagnostic methods,such as the use of chromogenic media with the ability to differentiate Candida species,as well asFinal version published online 6June 2011.M I C R O B I O L O G Y R E V I E W Sthe introduction of molecular techniques in the routine diagnosis of fungemia(Liguori et al.,2009).However,the high prevalence of NCAC species in disease could also be a reflection of their inherently higher level of resistance to certain antifungal drugs(Gonzalez et al.,2008)compared with C.albicans,as this would promote their persistence, possibly to the detriment of C.albicans,in mixed species infections treated with traditional antifungal agents. Unfortunately,compared with C.albicans there are relatively few studies examining the virulence factors of NCAC species.This review therefore provides information on the current state of knowledge on the biology,identifica-tion,epidemiology,pathogenicity and antifungal resistance of Candida glabrata,Candida parapsilosis and Candida tropicalis,three of the most frequent causes of candidosis after C.albicans.Biology of NCAC speciesCandida comprises an extremely heterogeneous group of fungal organisms that can all grow as yeast morphology. Macroscopically,colonies of Candida,on the routinely used Sabouraud dextrose agar(SDA),are cream to yellow in colour.Depending on the species,colony texture may be smooth,glistening or dry,or wrinkled and dull.Under standard conditions with optimal nutrients,yeast grow in log phase as budding cells(blastoconidia),which are sphe-rical to oval in shape and are approximately2–5Â3–7m m in size(Fig.1)(Larone,2002).Moreover,certain species,such as C.albicans and Candida dubliniensis,can produce a filamentous type of growth,such as true hyphae(Fig.1)or more frequently,pseudohyphae(Fig.1).The distinction between hyphae and pseudohyphae is related to the way in which they are formed.Pseudohyphae are formed from yeast cells or hyphae by budding(Fig.1), but the new growth remains attached to the parent cell and elongates,resulting infilaments with constrictions at the cell–cell junctions.There are no internal cross walls(septa) associated with pseudohyphae(Fig.1).In comparison,true hyphae are formed from yeast cells or even as branches of existing hyphae.The development of true hyphae is initiated by a‘germ tube’projection(Fig.1),which elongates and then branches with defined septa that divide the hyphae into separate fungal units(Fig.1).Candida albicans and C.dubliniensis are truly poly-morphic,due to their ability to form hyphae and/or pseudohyphae,and these species are also referred to germ tube positive,a diagnostic feature(Table1)(Calderone, 2002).In contrast,C.glabrata is not polymorphic,growing only as blastoconidia(yeast)(Table1;Fig.2).Historically, this species was originally classified in the genus Torulopsis due to its lack of pseudohyphal formation.However,in 1978,it was determined that the ability to form pseudohy-phae was not a reliable distinguishing factor for members of the genus Candida and it was proposed that Torulopsis glabrata could be classified in the genus Candida,due to its association with human infection(Fidel et al.,1999).With regard to C.parapsilosis,this species does not produce true hyphae,but can generate pseudohyphae that are character-istically large and curved,and often referred to as‘giantcells’Fig.1.Epifluorescence photocomposition of the different morphological growth forms of Candida albicans stained with calcofluor white:(A) blastoconidia;(B1)reproduction by budding;(B2)germ tube formation;(C1)pseudohyphae formation;(C2)yeast form;(C3)hyphae formation.ÃInternal cross walls(septa).Non-Candida albicans Candida species pathogenicity289(Fig.2)(Larone,2002;Trofa et al .,2008).In contrast,on corn meal Tween 80agar and at 251C after 72h,C.tropicalis produces oval blastospores,pseudohyphae depending on some reports,true hyphae (Fig.2)(Calderone,2002;Larone,2002;Yoshio &Kouji,2006).It should also be highlighted that C.glabrata cells (1–4m m)are noticeably smaller than the blastoconidia of C.albicans (4–6m m),C.tropicalis (4–8m m)and C.para-psilosis (2.5–4m m)(Larone,2002)(Table 1).On SDA (Fig.2)C.glabrata forms glistening,smooth,and cream-co-loured colonies,which are largely indistinguishable from those of other Candida species except for their relative size,which can be quite small (Fig.2).Furthermore,C.para-psilosis ,when grown on SDA,yields white,creamy,shiny and smooth/wrinkled colonies (Fig.2).On the same medium,C.tropicalis forms colonies that are cream-coloured with a slightly mycelial border (Fig.2)(Calderone,2002).Concerning the biochemistry of Candida species, C.glabrata ferments and assimilates only glucose and trehalose,which contrasts with C.albicans ,which ferments and/or assimilates a number of sugars with the notable exception of sucrose (Odds,1988).Additionally,C.tropicalis has the ability to ferment and assimilate sucrose and maltose(Martin,1979).Interestingly, C.parapsilosis was firstly classified as a species of Monilia ,due to its inability to ferment maltose (Odds,1988;Trofa et al .,2008).A main distinguishing genetic characteristic of C.glabrata is that it has a haploid genome,in contrast to the diploid genome of C.albicans and several other NCAC species (Fidel et al .,1999).Genetically, C.tropicalis has the highest similarity to C.albicans ,and C.glabrata the least (Butler et al .,2009).It is through the advent of molecular genetics that new identification methods for Candida have been developed,leading to the identification of new species together with their increased recognition in human infec-tion.Before 2005,C.parapsilosis was separated into three groups (I–III),but further studies revealing genomic differ-ences that have led to the separation of these groups into closely related,but distinct species,namely,C.parapsilosis ,Candida orthopsilosis and Candida metapsilosi s (Tavantiet al .,2005;G ´acser et al .,2007a,b).Laboratory identification of NCAC speciesThe laboratory diagnosis of candidoses continues to beproblematic.Microbiological confirmation can bedifficultFig.2.Candida species macroscopic colonies on cornmeal Tween 80and microscopy structure on SDA.Microscopic structures:(a)Candida glabrata ;(b)Candida parapsilosis ;(c)Candida tropicalis ;macroscopic colonies:(d)C.glabrata ;(e)C.parapsilosis ;(f)C.tropicalis .Table 1.Morphological characteristics of Candida albicans ,Candida tropicalis ,Candida parapsilosis and Candida glabrata species Species Germ tube production Hyphae/pseudohyphae Yeasts size (m m)CHROM-agar colony colour C.albicans 11/14–6Â6–10Blue-green C.tropicalis ÀÆ/14–8Â5–11Dark blue C.parapsilosis ÀÀ/1 2.5–4Â2.5–9WhiteC.glabrataÀÀ/À1–4White,Pink-purple290S.Silva et al.as blood cultures can be negative in up to50%of autopsy-proven cases of deep-seated candidoses,or may only become positive late in the infection(Ellepola&Morrison,2005). Positive cultures from urine or mucosal surfaces do not necessarily indicate invasive disease although may occur during systemic infection(Ellepola&Morrison,2005). Furthermore,differences in virulence between Candida species as well as in their susceptibility to antifungal drugs make identification important for clinical management. Laboratory diagnosis has improved with the advent of new methods for Candida isolation and identification. Technologies such as species-specific FISH(Alexander et al.,2006),antibody and antigen detection(Pfaller,1992; Ellepola&Morrison,2005)and molecular approaches for typing and detection of fungal pathogens(Ellepola& Morrison,2005)have all been used successfully.However, many of these approaches have not yet been standardized or validated in large clinical trials and therefore are not widely used in clinical laboratory settings(Ellepola&Morrison, 2005).Laboratory surveillance of‘at-risk’patients could result in earlier initiation of antifungal therapy if sensitive and specific diagnostic tests,which are also cost effective, become widely available.The clinical symptoms of fungemia are not indicative of particular Candida species and may be induced by other microorganisms.The laboratory identification of Candida is therefore essential for establishing a definitive diagnosis.A standard approach to the laboratory diagnosis generally involves nonmolecular methods,although PCR is increas-ingly being used.Non-PCR based methods of Candida identificationCHROMagar s Candida(CHROMagar s,Paris,France),is a relatively new differential agar medium for Candida species identification and has been particularly useful in the pre-sumptive identification of C.albicans, C.tropicalis and Candida krusei upon primary culture of clinical specimens. On CHROMagar s Candida,C.glabrata colonies appear white,pink or purple in contrast to C.albicans colonies, which are blue-green,while C.parapsilosis colonies are white and C.tropicalis dark blue(Table1).Moreover,it is possible to detect coinfection with different Candida species on primary culture plates and this can have importance in infection management strategies(Ellepola&Morrison, 2005;Furlaneto-Maia et al.,2007).After Candida isolation,species can also be identified by carbohydrate assimilation and fermentation tests as well as morphological characteristics such as germ tube and chla-mydospore development(Fig.2).In addition,more rapid and less laborious phenotypic identification methods have become available.Perhaps the most widely used methods for Candida species identification are those based on the format of carbohydrate assimilation and/or enzyme detection with-in plastic wells of commercially available kits.Examples of such biochemical tests include the API20C AUX(API Candida)Auxacolor(Bio-Rad)and the Uni-Yeast-Tek kit (Ellepola&Morrison,2005).These tests generate reliable identification for the most common species of Candida, while identification of other Candida species may not be so accurate.For example,the differentiation of C.dubliniensis from C.albicans often requires the use of supplemental biochemical or morphological tests for definitive identifica-tion(Verweij et al.,1999;Ellepola&Morrison,2005). Additional methods for Candida species identification include tests that allow the detection of an isolate in1day, such as the RapID Yeast Plus System(Innovative Diagnostic Systems,Norcross),the Fongiscreen test(SanofiDiagnostics Pasteur,France)and the automated Rapid Yeast Identifica-tion Panel(Dade Microscan).However,as mentioned above,most of these tests tend to be most accurate for the identification of the more frequently encountered yeast pathogens(Ellepola&Morrison,2005).The diagnosis of invasive candidosis should include a collection of adequate volumes of blood and an agar-based blood culture method for optimal detection of candidemia (Pfaller,1992).Several advances in blood culturing techni-ques have been developed,which appear to have improved the sensitivity and/or reduced the time required to obtain a positive blood culture.Two automated methods for mon-itoring of blood culture bottles,based on colour(BacT/ ALERT3D,Organon Teknika Corp.,Durham,NC)and fluorescence(BACTEC9240,Becton Dickinson),have been developed recently(Ellepola&Morrison,2005).The identification of typical blastospores and pseudohy-phae of Candida species on microscopic examination of tissue remains the unequivocal standard for the diagnosis of invasive or disseminated candidoses.Unfortunately,the usefulness of this approach is frequently limited by sampling problems(isolation source and sample size)(Pfaller,1992). The use offluorescent antibody,acridine orange or calco-fluor-white staining(Pfaller,1992;Ellepola&Morrison, 2005)may enhance the sensitivity of microscopic examina-tion.However,the production offluorescent antibodies specific for the identification of individual Candida species has proved to be extremely difficult.A relatively recent laboratory method based on PNA FISH targeting the26S rRNA gene allows the reliable detection of C. albicans from NCAC species,within2.5h of yeast growth detection in blood culture,with a sensitivity of99%and specificity of100%(Rigby et al.,2002).According to recent studies PNA FISH also results in substantial cost savings for hospitals,making the method both an effective and affordable one for the laboratory diagnosis of candidoses(Rigby et al., 2002;Ellepola&Morrison,2005;Alexander et al.,2006).Non-Candida albicans Candida species pathogenicity291PCR-based methods of Candida identification The molecular-based technology that has undoubtedly had the greatest impact in the clinical diagnosis of Candida infections is PCR.This technique can detect highly limited quantities of microbial nucleic acid from blood,tissue specimens as well as cultured microorganisms.Over the last decade,numerous studies have been performed to investi-gate the effectiveness of PCR in diagnosis of systemic infection caused by Candida(Williams et al.,1995,2001; Chen et al.,2000;Carvalho et al.,2007;Orazio et al.,2009). In PCR,a pair of synthetic oligonucleotides homologous to specific sequences serves to prime the amplification of target DNA.The most important feature of any PCR primers used directly on clinical samples is that they are specific and do not amplify host DNA or that of other microorganisms.To improve the sensitivity of PCR,many investigators have designed primers that amplify regions of DNA that are repeated in the fungal genome.The most commonly used target for yeast diagnostic PCR primers is the rRNA gene operon,encoding the18S, 5.8S,and28S rRNA gene subunits,namely internal transcribed spacer1(ITS1),ITS2 and ITS4(Fell et al.,1992;Sullivan et al.,1995;Williams et al.,1995,2001;Haynes&Westerneng,1996;Chen et al., 2000).More recently,multiplex targets,coupled to real-time PCR,have been used successfully(Sampaio et al.,2005; Carvalho et al.,2007;Orazio et al.,2009)for Candida species identification.Despite the increased development of new molecular approaches,the great majority of clinical diagnosis of candidosis are based on nonmolecular methodologies due the reduced amount of PCR equipment in hospital labora-tories,the problems with sample preparation and environ-mental contamination and the lack of standardized protocols for PCR methodologies.Epidemiology and risk factors in NCAC species infectionThe mortality rates associated with different microorgan-isms have declined with the early administration of empiri-cal antibiotics and antifungal agents.However,despite this, systemic fungal infections are increasingly recognized as important causes of morbidity and mortality.Candida species are among the most frequently recovered fungi from blood cultures of hospitalized patients(Pfaller et al.,1998, 2010).In fact,an increasing incidence of fungal infections with Candida species has been noted in immunocompro-mised patients,including those in intensive care,postsurgi-cal units and suffering from cancer(Kiehn et al.,1980; Samaranayake et al.,2002;Hagerty et al.,2003).Candida species are most frequently isolated from the oral cavity,and vulvovaginal and urinary tracts and are detected in approxi-mately31–55%of healthy individuals.Historically, C. albicans has accounted for70–80%of clinical isolates,with other NCAC species only rarely encountered(Odds,1988; Calderone,2002;Samaranayake et al.,2002).Nevertheless, over the last10–30years NCAC species have emerged as important opportunistic pathogens of humans and the reasons for this might be related to improved diagnostic methods or altered medical practices,as mentioned above. Regardless of the basis of this change,recent epidemiological data reveal a mycological shift,and while C.albicans remains the most common causative agent,its relative incidence in infection is declining with the increasing prevalence of other species such as C.glabrata,C.tropicalis and C.parapsilosis (Chandra et al.,2001;Colombo et al.,2003;Bassetti et al., 2006).In a study on the epidemiology of invasive candidosis, Pfaller&Diekema(2007)observed that C.albicans,C. glabrata,C.tropicalis and C.parapsilosis collectively ac-counted for about95%of identifiable Candida infections. Moreover,in the1980s,according to Kiehn et al.(1980),C. albicans constituted68%of Candida isolates from sites other than blood in cancer patients,while C.tropicalis,C. parapsilosis and C.glabrata accounted only for12%,10% and 3.0%of the isolates,respectively.Table2presents epidemiologic studies published between2000and2010, concerning oral candidosis,candiduria and candidemia.In more recent studies,most cases of fungemia have been significantly associated with NCAC species(Bassetti et al., 2006;Colombo et al.,2007;Chakrabarti et al.,2009;Pfaller et al.,2010).However,it is important to emphasize that there are significant variations in Candida species isolation depending on the geographical region and patient group, with some NCAC species being more prevalent,even compared with C.albicans,in certain countries(Colombo et al.,2007).The incidence of C.glabrata is higher in adults than in children,and lower in neonates(Krcmery&Barnes,2002). In contrast, C.parapsilosis appears to be a significant problem in neonates,transplant recipients and patients receiving parenteral nutrition(Trofa et al.,2008).Further-more,C.tropicalis is commonly associated with patients with neutropenia and malignancy(Colombo et al.,2007). For many years C.glabrata was considered a relatively nonpathogenic saprophyte of the normalflora of healthy individuals and certainly not readily associated with serious infection in humans.However,following the widespread and the increased use of immunosuppressive therapies together with broad-spectrum antibiotic treatment,the frequency of mucosal and systemic infections caused by C.glabrata has increased significantly(Hajjeh et al.,2004). Although the mortality rate associated with Candida infec-tions varies with the type of patient and with the causative agent,the incidence rates of candidosis infections attributed292S.Silva et al.to NCAC species were14%for C.glabrata and C.parapsilosis and7%for C.tropicalis according to a European Confedera-tion of Medical Mycology survey(Tortorano et al.,2006). Recently,Chen et al.(2008)reported that C.glabrata was a causative agent of candiduria in Australia.This is extremely important,because,compared with other NCAC species infection,the mortality rate associated with C.glabrata is the highest(Abi-Said et al.,1997;Krcmery,1999b).Until recently,few studies had evaluated independent risk factors associated with nosocomial C.glabrata acquisition and subsequent disease.Although C.glabrata is known to be present in patient’sflora,relatively little is known about the hospital reservoirs of C.glabrata,with likely sources of infection involving a complex interaction of both environ-mental and human reservoirs.Two studies(Isenberg et al., 1989;Vazquez et al.,1993)have indicated hand carriage on hospital personnel as possible sources of infection.Thus, similar to other nosocomial pathogens,C.glabrata may also be acquired,directly or indirectly,from contaminated environmental surfaces.However,the role of carriage by personnel in dissemination of C.glabrata remains to be clarifitely,the most frequent combination of mixed species infection by Candida species is C.glabrata and C.albicans,which has been found in approximately70%of the patients with oral candidosis(Redding et al.,2002).Candida parapsilosis,despite being initially considered a nonpathogenic species,was initially identified as the causa-tive agent of a fatal case of endocarditis in an intravenous drug user in1940(Joachim&Polayes,1940).Furthermore, over the past decade,the incidence of C.parapsilosis in infections has increased drastically.In fact,reports indicate that C.parapsilosis is often the second most frequently isolated Candida species from blood cultures(Almirante et al.,2006;Colombo et al.,2007;Costa-de-Oliveira et al., 2008).Furthermore,C.parapsilosis is one of the fungi most frequently isolated from human hands(Bonassoli et al., 2005)and the second most commonly isolated Candida species from normally sterile body sites of hospitalized patients.This species accounts for15.5%of Candida isolates in North America,16.3%in Europe,and23.4%in Latin America,outranked only by C.albicans(51.5%,47.8%and 36.5%,respectively)and C.glabrata(21.3%)in North America(Messer et al.,2006).However, C.parapsilosis fungemia has a lower mortality rate(4%)compared with that caused by C.albicans and C.glabrata(Kossoff et al., 1998).The increased incidence of C.parapsilosis infections has been attributed to a variety of risk factors,similar to other Candida species,including the organism’s selective growth capabilities in hyperalimentation solutions and its highTable2.Selected epidemiological studies published from2000to2010,concerning the distribution of Candida species isolates among various types of candidosisCandidosis References Period ofobservationRegion/countryNumberof strainsC.albicans(%)C.tropicalis(%)C.parapsilosis(%)C.glabrata(%)Oral candidosis Gonzalez Gravina et al.(2007)February–May2003Venezuela4342.312.814.9 2.1Martins et al.(2010a,b)May2005–2006Portugal5379 4.8 6.5 4.8Luque et al.(2009)-Argentine-60.7 4.5- 5.6 Candiduria Kauffman et al.(2000)-USA53051.87.9 4.115.6 Kobayashi et al.(2004)-Brazil4535.522.311.18.8Passos et al.(2005)-Brazil4370 4.6 4.67Binelli et al.(2006)1999–2001Brazil235243.5-17.3Chen et al.(2008)June–August2006Australia6585.2- 4.427.8A´lvarez-Lerma et al.(2003)1998–1999Spain38968.4360.58.2Dorko et al.(2002)-Slovakia9461.7 6.324.5-Candidemia Hazen et al.(1986)-USA1262138123 Chakrabarti et al.(2009)-India-26.3-10.5Colombo et al.(2007)-Brazil2823848239Costa-de-Oliveira et al.(2008)During2004Portugal-35-26.5-Bassetti et al.(2006)1999–2003Italy1824092315Miranda et al.(2009)2004–2005Brazil-4233162Tortorano et al.(2006)1997–1999Europe4735371414Trick et al.(2002)During1999USA-59101112Pfaller et al.(2010)2008–2009Europe/Asia/American1239509.817.417.4-,Not mentioned.Non-Candida albicans Candida species pathogenicity293ability to colonize intravascular devices and prosthetic materials.Additionally,patients requiring prolonged used of a central venous catheter or indwelling devices,such as cancer patients,are at increased risk of infection with C.parapsilosis.A recent Spanish study of72patients with invasive C.parapsilosis identified vascular catheterization (97%),prior antibiotic therapy(91%),parenteral nutrition (54%),prior surgery(46%),prior immunosuppressive therapy(38%),malignancy(27%),transplant receipt (16%),neutropenia(12%)and prior colonization(11%), as risk factors for infection(Almirante et al.,2006).In a report of64episodes(between2002and2003)of C.parapsilosis candidemia in Brazilian hospitals,the pri-mary risk factors were neutropenia,the use of central venous catheters and cancer chemotherapy(Brito et al.,2006).The population at greatest risk for nosocomial infection with C.parapsilosis is that of extremely low-birth-weight neonates (Solomon et al.,1984;V oss et al.,1994).In fact,colonization of the skin or gastrointestinal tract is frequently thefirst step in the pathogenesis of invasive candidosis,and neonates are especially prone to such infections given their compromised skin integrity,susceptibility to gastrointestinal tract infection, long-term need for central venous or umbilical catheters and prolonged endotracheal intubation(Benjamin et al.,2000). Furthermore,C.parapsilosis has been isolated from approxi-mately one-third of neonates with gastrointestinal coloniza-tion by Candida species(Saiman et al.,2001)and from oropharynges of23%of healthy neonates(Contreras et al., 1994).Furthermore,in contrast to other NCAC species,the rates of mortality in low-birth-weight neonates caused by C.parapsilosis are drastically higher and sometimes equivalent to those associated with C.albicans(Trofa et al.,2008). Candida tropicalis is one of the three most commonly isolated NCAC species(A´lvarez-Lerma et al.,2003;Binelli et al.,2006;Colombo et al.,2007;Hasan et al.,2009). Usually,C.tropicalis is considered the third most frequently isolated NCAC species from blood and urine cultures(Table 2)(Kauffman et al.,2000;A´lvarez-Lerma et al.,2003). Moreover,in a recent epidemiology study conducted in12 Brazilian medical centres,C.tropicalis was the second most frequently recovered Candida species,accounting for 33–48%of all candidemia cases(Colombo et al.,2007; Miranda et al.,2009).Additionally,C.tropicalis is often found in patients admitted to intensive care units,especially in patients requiring prolonged catheterization,receiving broad-spectrum antibiotics or with cancer(Kauffman et al., 2000;Rho et al.,2004;Colombo et al.,2007;Nucci& Colombo,2007).Furthermore,C.tropicalis appears to dis-play a higher potential for dissemination in neutropenic individuals compared with C.albicans and other NCAC species(Colombo et al.,2007).According to Kontoyiannis et al.(2001),there are distinct differences in the presentation and risk factors of C.tropica-lis and C.albicans fungemia,with the former more persis-tent and leading to longer intensive care unit stays during the course of infection.This may imply a higher virulence and greater resistance to commonly used antifungals by C.tropicalis when compared with C.albicans.In fact,some epidemiologic studies(Krcmery,1999a;Kontoyiannis et al., 2001;Eggimann et al.,2003;Colombo et al.,2007) documented that C.tropicalis was associated with higher mortality than other NCAC species and C.albicans. This propensity of C.tropicalis for dissemination and the associated high mortality may be related to the virulence factors exhibited by this species such as biofilm formation, proteinases secretion and dimorphism(Krcmery,1999b; Negri et al.,2010a).Pathogenicity and virulence factors of NCAC speciesThere remains a debate over what actually constitutes a virulence factor.It can be argued that all the traits required for establishing disease are virulence factors;however, strictly speaking,virulence factors are those that interact directly with host cells causing damage(Haynes,2001).The pathogenicity of Candida species is mediated by a number of virulence factors,including adherence and biofilm forma-tion on host tissue as well as medical devices,the ability to evade host defences and the production of tissue-damaging hydrolytic enzymes(e.g.proteases,phospholipases and haemolysins).Infection models of candidosis in animals suggest that C.albicans is the most pathogenic species(Samaranayake& Samaranayake,2001),and in vitro investigations indicate that it also expresses higher levels of putative virulence factors compared with other species(Jayatilake et al., 2006).Furthermore,it is important to emphasize that these yeasts are not usual pathogens of these animals and therefore such studies do not necessarily reflect the reality of patho-genicity of Candida species.Moreover,Candida species can colonize and cause disease at several anatomically distinct sites including the skin,oral cavity,gastrointestinal tract, vagina and vascular system.In order to establish infection, opportunistic pathogens have to evade the immune system, survive,reproduce in the host environment,and in the case of systemic infection,disseminate to new tissues and organs. Adhesion and biofilm formationThe primary event in Candida infection is adherence to host surfaces,which is required for initial colonization.Adher-ence contributes to persistence of the organism within the host,and is considered essential in the establishment of disease.Furthermore,Candida species can also adhere to the surfaces of medical devices and form biofilms.Several factors have been implicated in influencing adhesion,294S.Silva et al.。
钱洗谦,乔乐克,张洪锋,等. 海洋细菌Sphingomonas sp. Q2产琼胶酶发酵条件优化、酶学性质及降解产物研究[J]. 食品工业科技,2023,44(18):139−146. doi: 10.13386/j.issn1002-0306.2022090058QIAN Xiqian, QIAO Leke, ZHANG Hongfeng, et al. Optimization of Fermentation Conditions, Enzymatic Properties and Degradation Products of Agarase Produced by Marine Bacterium Sphingomonas sp. Q2[J]. Science and Technology of Food Industry, 2023, 44(18):139−146. (in Chinese with English abstract). doi: 10.13386/j.issn1002-0306.2022090058· 生物工程 ·海洋细菌Sphingomonas sp. Q2产琼胶酶发酵条件优化、酶学性质及降解产物研究钱洗谦1,乔乐克2,张洪锋3,江晓路4,王 鹏1,张京良2,4, *(1.中国海洋大学食品科学与工程学院,山东青岛 266003;2.青岛海洋生物医药研究院,山东青岛 266100;3.青岛海莱美生物科技有限公司,山东青岛 266400;4.中国海洋大学医药学院,山东青岛 266003)摘 要:本研究旨在对从江蓠中筛选获得的Sphingomonas sp. Q2菌株产琼胶酶能力条件进行优化并对其酶学性质及降解产物进行研究。
通过响应面法对发酵条件进行优化,采用硫酸铵沉淀、离子交换层析和凝胶层析等方法对发酵所得酶液进行纯化并对纯化后的酶液进行酶学性质研究。
Quantabio, 100 Cummings Center Suite 407J, Beverly, MA 01915IFU-115.1 Rev01repliQa™ HiFi Assembly MixCat. No. 95190-010 95190-050Size:10 reactions 50 reactionsStore at -25°C to -15°CDescriptionThe repliQa™ HiFi Assembly Mix simplifies the construction of recombinant DNA through the simultaneous and seamless assembly of multiple DNA fragments possessing terminal regions of sequence overlap in a single, isothermal reaction. Similar in principle to the Gibson Assembly ® Method 1, the high efficiency repliQa HiFi Assembly Mix is ideal for a range of genetic engineering applications including routine molecular cloning, site-directed mutagenesis, assembly of large constructs for synthetic biology applications, and the construction of diverse sequence libraries for directed evolution studies. The concentrated, two-component format allows flexibility in design of assembly reactions and compatibility with less concentrated DNAsamples. The repliQa Mix has been optimized for use with a total input quantity of DNA fragments in the range of 0.03 to 0.5 pmols. The assembly of up to six DNA fragments is recommended, though the repliQa Mix has been successfully used for more complex assemblies.Double stranded DNA fragments for assembly can be generated by PCR amplification, chemical synthesis, or isolation of restriction fragments. When working with fragments PCR amplified from plasmid vectors, the included DpnI restriction endonuclease can be used for selectively digesting methylated, residual plasmid DNA to reduce background transformants. The repliQa mix is directly compatible with most common E. coli cloning hosts and generally provides a high yield of accurately assembled product.The DNA assembly occurs through the actions of three enzymes:• A non-thermostable 5' to 3' exonuclease that partially eliminates one strand of a DNA duplex to expose complementary overlap regions forhybridization.• A high-fidelity thermostable polymerase that fills the gaps remaining between the hybridized fragments of the overlapping regions.• A thermostable DNA ligase that covalently seals the resulting nicks at fragment junctions, generating double-stranded, assembled DNA moleculessuitable for transformation of cells.ComponentsReagent Description95190-01095190-050 repliQa HiFi Assembly Enzyme Mix Optimized formulation of enzymes for 5’-endresection, high fidelity 3’-end extension, and nick sealing.1 x 0.02 mL1 x 0.10 mLrepliQa 10X Assembly Reaction Buffer 10X reaction buffer containing dNTPs, magnesium, and cofactors.1 x 0.1 mL 1 x 0.50 mLDpnI (20 U/µl)Restriction endonuclease for the (optional) post-PCR digestion of residual unamplified plasmid template.1 x 0.05 ml 1 x 0.25 mlStorage and StabilityStore kit components in a constant temperature freezer at -25°C to -15°C upon receipt. For long term buffer storage (> 30 days) store buffer at -70°C. Refer to the product label or lot-specific Product Specification Sheet (PSF) available at /resources for applicable expiration date.A general diagram of assembly cloning is shown below:Additional reagents and materials that are not supplied• PCR-Grade, nuclease-free water (do not use DEPC-treated water)• High Fidelity DNA Polymerase (Enzymatics VeraSeq TM 2.0, P7511L or equivalent)• A heat block, thermocycler, or water bath capable of holding a temperature of 50 ± 2°C for one hour. • PCR or microcentrifuge reaction tubes.• PCR product purification kit (QIAGEN ® QIAquick ® PCR Purification Kit, 28104 or equivalent). •Competent E. coli cells and accessories as recommended by manufacturer.Before you begin• Design the DNA fragment sequences and assembly strategy. Guidelines are given in Appendix 1.• (Optional) Treat PCR reaction with DpnI if plasmid DNA was used as template for generating DNA fragments to be assembled. (Appendix 2).•(Recommended) After determining PCR fragment or restriction endonuclease-digested fragment size and purity by agarose gel electrophoresis, purify using a spin column-based cleanup or other method. This step is not required but is highly recommended to achieve highest efficiency of fragment assembly.• Measure the concentration of each isolated DNA fragment by absorbance at A 260 or by using a fluorometric quantitation reagent. Agarose gel electrophoresis with mass-calibrated size standards can also be used to quantify fragment mass and quality simultaneously. • Calculate the number of picomoles of each fragment using the following formula:pmols = (weight in ng) x 1000/(bp x 662).• Determine the number of pmols of each fragment to add to the assembly reaction. For cloning, highest efficiencies are achieved with 0.02 to 0.04 pmols of linear vector fragment (50 to 100 ng of 4 kb vector) and 2 to 8-fold molar excess of inserts. • Prepare outgrowth medium and culture plates with appropriate antibiotics for plasmid selection.•Equilibrate the heat block, thermal cycler, or water bath to 50°C for incubation of the assembly reactions .Protocol1. Thaw the repliQa HiFi Assembly Kit components, briefly vortex to mix, and place on ice.2. For each assembly, add reaction components in the order listed in the table below to chilled reaction tubes.The optimal amount of enzyme mix to add per assembly reaction depends on the total quantity of DNA fragments present.ComponentRxn. component volumes (µl) for varying amounts of total DNA≤ 0.125 pmol> 0.125 pmol but ≤ 0.25 pmol > 0.25 pmol Nuclease-free water(17.5 – X) µl (17.0 – X) µl (16.0 – X) µl repliQa 10X Assembly Reaction Buffer 2.0 µl 2.0 µl 2.0 µl DNA fragmentsX µl X µl X µl repliQa HiFi Assembly Enzyme Mix 0.5 µl 1.0 µl 2.0 µl Total volume20 µl20 µl20 µl3. Incubate reactions at 50°C in heat block, thermal cycler with heated lid (set to ~60-80°C), or covered water bath for 1 hr. Hold assembled product mix at 4°C until ready to proceed with transformations. If transformations cannot be performed on the same day, reactions can be stored at -20°C for up to one month.4. Competent E. coli should be transformed, recovered, and plated as per manufacturer guidelines or standard lab practices. Note: If electroporation is to be used for transforming cells, we recommend first diluting the assembly reaction 1:5 in high purity water. There is no need to dilute the assembly reactions prior to transformation of chemically competent cells.5. (Optional) Analyze a portion of the remaining assembly reaction by agarose gel electrophoresis. If DNA fragment assembly occurs properly, a ladder of higher molecular weight DNA bands would be generated.Note: For reactions using three or fewer fragments the incubation time in step 3 can be shortened to 15 minutes.Appendix 1 – Guidelines for Designing DNA Fragments for Assembly1.When designing the DNA fragment sequences and assembly strategy, allow for a region of sequence homology between adjacent DNA fragments.Be sure to avoid regions of repeated bases or repeated short DNA motifs in the design of these overlaps where possible. Regions of secondary structure such as hairpins or stem loops should also be avoided.2.The kit is optimized for the assembly of fragments with overlap regions between 15 – 60 bp. It is recommended that the overlaps be at least 20bp with a minimum of 25% GC content, however overlaps of 30 bp or longer size will provide higher efficiency assembly reactions.3.For generating PCR fragments to be assembled, design primers with a 5’ segment of homology to the adjacent fragment or vector. If the adjacentfragment is also generated by PCR amplification, the overlap can be split between two primers if desired. The 3’ segment of primers should contain sequence specific to the DNA target of interest. Amplify targets using a high-fidelity thermostable DNA polymerase such as VeraSeq 2.0 (Enzymatics, P7511L) or equivalent per manufacturer instructions.4.When designing synthetic gene fragments for assembly, ensure that the 5’ and 3’ segments contain regions of homologous overlap sequencebetween adjacent gene blocks, PCR fragments, or isolated restriction fragments.5.For site-directed mutagenesis applications, the assembly strategy should be designed such that the mutation of interest is centered betweenadjacent PCR fragments. Design the PCR primers as with the standard fragments above, except that the mutation (substitution, insertion, or deletion) should be included within the 5’ segments for both of the adjacent fragments.6.When designing DNA fragments to be assembled with isolated restriction fragments, be aware that any 5’ overlaps from staggered restriction cutswill be eliminated because of the 5’-->3’ nuclease present in the assembly mix, and so should not be included in the measurement of overlap size. If desired, design the 5’ overlap segment of the adjacent fragment to either preserve or eliminate the restriction site.Appendix 2 – DpnI treatment to remove residual plasmid DNAWhen plasmid vector is used as PCR template to generate a fragment for assembly, it is recommended that the reaction be treated with DpnI to eliminate residual methylated plasmid prior to setting up the assembly reaction.1.Add 1 µl DpnI (20U) directly to the PCR reaction (50 µl) following amplification of fragment.2.Incubate at 37°C for 1 hr.3.Heat inactivate DpnI by incubation at 80°C for 20 min.4.(Recommended) Purify the fragment using a spin column-based PCR purification kit.Quality ControlThe repliQa HiFi Assembly Mix is functionally tested for assembly of three 1-kb PCR fragments into 2kb and 3 kb products.The individual components of the repliQa HiFi Assembly Mix are tested to be free of contaminating DNase and RNase.Limited Label LicensesThis product was developed, manufactured, and sold for in vitro use only. The product is not suitable for administration to humans or animals. SDS sheets relevant to this product are available upon request.References1. Gibson, D.G., et al. (2009). Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods 6, 343-5.。
专利名称:COMPOSITIONS CONTAININGCOMBINATIONS OF NITROGEN-FIXINGBACTERIA AND ADDITIONAL AGENTS ANDTHEIR USE IN FIXING NITROGEN IN PLANTSPECIES发明人:AVIDOV, Amit,BARAZANI, Avner,ZEILKHA, Mor申请号:IL2019/050082申请日:20190121公开号:WO2019/142199A1公开日:20190725专利内容由知识产权出版社提供专利附图:摘要:The present invention provides a method for supplying the nitrogen requirements of a plant comprising administering to said plant a combination of non-pathogenic, atmospheric nitrogen-fixing bacteria and one or more activating agents. Many of these activating agents possess potent anti-inflammatory and anti-microbial activity. The method is particularly suitable for use in enabling nitrogen fixation in plant species such as wheat, maize and other cereal crops, in which nitrogen fixation is normally not possible. The invention also provides compositions comprising nitrogen-fixing bacteria and suitable activating agents. In one preferred embodiment, the nitrogen fixing bacteria are of the genus.申请人:GRACE BREEDING NITROGEN FIXATION TECHNOLOGIES LTD.地址:11B, Reik Haviva Street 4428327 Kfar Saba IL国籍:IL代理人:RUTMAN, Avraham更多信息请下载全文后查看。
专利名称:MESENCHYMAL STEM CELLS WITH ENHANCED EFFICACY发明人:Neil RIORDAN申请号:US16133581申请日:20180917公开号:US20190030081A1公开日:20190131专利内容由知识产权出版社提供摘要:Disclosed are protocols, isolation means, and compositions of matter useful for identifying mesenchymal stem cells possessing enhanced clinical activity. In one embodiment, markers associated with said enhanced mesenchymal stem cell activity are utilized to identify donors whose mesenchymal stem cells possess superior efficacy compared to mesenchymal stem cells from donors who lack said markers associated with said enhanced efficacy. In one embodiment, said markers are utilized to select for mesenchymal stem cells possessing enhanced efficacy from in vitro cultures. In another embodiment, surfaces markers associated with said markers associated with enhanced efficacy are utilized to positively select for cells possessing enhanced efficacy. In another embodiment, the invention teaches markers whose expression is correlated with negative efficacy. Said markers can be utilized to exclude mesenchymal stem cell donors, or in vitro generated and/or isolated mesenchymal stem cells prior to clinical use. In another embodiment the invention teaches a method of augmenting mesenchymal stem cell efficacy by inhibiting the expression of proteins found in higher concentrations in cells without enhanced clinical activity. Additionally, novel mesenchymal stem cells phenotypes are disclosed possessing enhanced efficacy compared to existing mesenchymal stemcells based on unique phenotypic characteristics.申请人:CELL MEDICINE, INC.地址:Farmers Branch TX US国籍:US更多信息请下载全文后查看。
专利名称:Isolated converter发明人:Koji Nakajima,Shota Sato,Yuji Shirakata,KentaFujii,Jun Tahara申请号:US16310667申请日:20170310公开号:US11239021B2公开日:20220201专利内容由知识产权出版社提供专利附图:摘要:An isolated converter reduced in size compared with a conventional isolated converter and having a high heat dissipation characteristic is provided. The isolatedconverter includes a multilayer substrate having a first through hole and a magnetic corepartially passing through the first through hole. The multilayer substrate includes a first conductor pattern formed at a position overlapping the magnetic core on a second surface when viewed from a direction orthogonal to a first surface, a second conductor pattern formed between the first surface and the second surface at a position overlapping the magnetic core and the first conductor pattern when viewed from the direction orthogonal to the first surface, at least one thermal conductive member formed on the first conductor pattern and having a portion disposed between the multilayer substrate and the magnetic core, and an electric insulating layer electrically insulating the first conductor pattern from the second conductor pattern.申请人:Mitsubishi Electric Corporation地址:Tokyo JP国籍:JP代理机构:Buchanan Ingersoll & Rooney PC更多信息请下载全文后查看。
a r X i v :a s t r o -p h /0211341v 2 21 N o v 2002Astrophysical Journal Letters,in press (scheduled Dec.1,2002)Preprint typeset using L A T E X style emulateapj v.20/04/00ISOLATED STAR FORMATION:A COMPACT HII REGION IN THE VIRGO CLUSTER 1Ortwin Gerhard 1,Magda Arnaboldi 2,3,Kenneth C.Freeman 4,Sadanori Okamura 51Astronomisches Institut,Universit¨a t Basel,Venusstrasse 7,CH-4102Binningen,Switzerland2I.N.A.F.,Osservatorio Astronomico di Capodimonte,80131Naples,Italy 3I.N.A.F.,Osservatorio Astronomico di Pino Torinese,10025Pino Torinese,Italy4R.S.A.A.,Mt.Stromlo Observatory,2611ACT,Australia5Department of Astronomy,School of Science,University of Tokyo,Tokyo,113-0033,JapanAstrophysical Journal Letters,in press (scheduled Dec.1,2002)ABSTRACTWe report on the discovery of an isolated,compact HII region in the Virgo cluster.The object is located in the diffuse outer halo of NGC 4388,or could possibly be in intracluster space.Star formation can thus take place far outside the main star forming regions of galaxies.This object is powered by a small starburst with an estimated mass of ∼400M ⊙and age of ∼3Myr.From a total sample of 17HII region candidates,the present rate of isolated star formation estimated in our Virgo field is small,∼10−6M ⊙arcmin −2yr −1.However,this mode of star formation might have been more important at higher redshifts and be responsible for a fraction of the observed intracluster stars and total cluster metal production.This object is relevant also for distance determinations with the planetary nebula luminosity function from emission line surveys,for high-velocity clouds and the in situ origin of B stars in the Galactic halo,and for local enrichment of the intracluster gas by Type II supernovae.Subject headings:stars:formation –HII regions –galaxies:ISM –galaxies:star clusters –galaxies:abundances –intergalactic medium1.INTRODUCTIONStars are usually observed to form in galaxies,that is,in disks,dwarfs,and starbursts.In radio galaxies,star formation may be triggered by energetic jet outflows (e.g.,Bicknell et al.2000).The HII region we describe here shows that isolated star formation takes place in the dif-fuse outskirts of galaxies,at the boundary of,if not already in,Virgo intracluster space.In nearby galaxy clusters,a diffuse intracluster star com-ponent has been inferred from surface brightness measure-ments (Bernstein et al.1995)and detection of individual stars (e.g.,Arnaboldi et al.1996,Ferguson et al.1998,Feldmeier 2002).Its origin may be explained readily by dynamical processes acting on low-surface brightness disks and dwarfs,which unbind the stars from these galaxies (Moore et al.1999).However,gas may be efficiently re-moved from infalling galaxies by ram pressure stripping (Quilis et al.2000,Gavazzi et al.2001),or may be tidally dissolved,or could fall into the cluster as pristine clouds.Some of this gas may form stars,which would also con-tribute to the diffuse component.We discuss here the current rate of such star formation from isolated compact HII regions (ICHIIs)and from jet induced star formation in a Virgo cluster field.2.AN HII REGION IN VIRGO:OBSERVED PROPERTIESThe target object was found in an emission line survey for planetary nebulae in a Virgo intracluster field,centred at α(J 2000)12:25:31.9,δ(J 2000)12:43:47.7,using H αand [OIII]narrow band and V+R broad band filterphotometry (Arnaboldi et al.2002,Okamura et al.2002),with Suprime-Cam on the Subaru Telescope.Because of the large H αto [OIII]flux ratio the (unresolved)object was classified as a candidate compact HII region.A spectrum was taken at UT4of the VLT at Paranal,on the night of April 14,2002,with FORS2in MOS mode.The observations were carried out with GRISM-150I and the order separation filter GG435+81,giving a wavelength coverage of 4500−10200˚A and a dispersion of 6.7˚A pix −1.The slit width was 1.4arcsec,and the angular scale along the slitlet was 0.126arcsec pix −1.The total exposure time was 7×1800s .The nights were clear but not photometric;the mean seeing was better than 1.0arcsec.Spectrophoto-metric standard stars were observed at the beginning and end of the night,but cirrus clouds at these times made the flux calibration uncertain.The data reduction was carried out using standard tasks in IRAF,using an arc lamp wavelength calibration and observations of a spectrophotometric standard star.The spectrum was corrected for atmospheric extinction,using a table for La Silla 2.The wavelength and flux-calibrated spectrum is shown in Figure 1:it clearly shows a blue continuum and a number of emission lines at Virgo redshift:H α,H β,H γ,[OIII]λ5007and λ4959,[SII]λλ6717+6731,and [SIII]λ9069.We do not resolve H αand [NII]λ6548,and the [SII]λλ6717,6731lines,respectively,and we do not see the weaker [OI]λ6300,[OIII]λ4363,and [SIII]λ6312lines.The H γline lies close to the blue end of the spectrum,and the [SIII]λ9069line in a region of strong sky emission.1Based on observations carried out at UT4of the VLT,Paranal,Chile,which is operated by the European Southern Observatory.2Nocorrection for atmospheric refraction is needed since FORS2has an atmospheric dispersion corrector.12Gerhard et al.500060007000800090002468Wavelength (angstroms)Fig.1.—Observed emission spectrum.Table 1Observed and reddening-corrected emission line fluxes relative to H β.H γ43400.580.71H β4861 1.00 1.00[OIII]4959 1.030.99[OIII]5007 3.05 2.91[NI]52000.08H α6563 4.30 2.89[NII]65830.750.50[SII]6717+67310.400.26[SIII]90690.300.14Isolated HII Region in Virgo Cluster3The observed emission lines and their uncorrected and reddening-correctedfluxes normalized to Hβare listed in Table1.The errors in thefluxes are approximately10% of the Hβflux,and larger at the blue edge of the spectral range.The Hαand[NII]λ6548lines are unresolved,with the[NII]λ6583line appearing in the red wing of Hα.From a two-Gaussianfit to the combined emission we estimate the line ratio[NII]λ6583/(Hα+[NII]λ6548)≃0.17.This is consistent with the value expected for an HII region with the large observed[OIII]λ5007/Hβratio;see below.The Hαflux given in the table is that measured as the totalflux in the line reduced by one third that in[NII]λ6583.The observed Balmer decrement Hα/Hβthen becomes4.3.If the intrinsic Balmer decrement has the theoretical value of2.89for large optical depth(case B)and temper-ature T=104K,then E(B-V)=0.40and A V=1.23mag. Most of this reddening is intrinsic to the source,as the galactic absorption to nearby galaxies in the Virgo cluster is about A V≃0.12.Linefluxes were corrected for redden-ing using this value of E(B-V)and the reddening curve of Cardelli et al.(1989).Compared to Hβ,the corrected Hγflux is about1.5times larger than expected;however,we cannot regard the Hγflux as reliable,because the line lies right at the edge of the spectrum where uncertainties in the grism response,sky subtraction and determination of the continuum level will be at their worst.The corrected line ratios[OIII]λ5007/Hβ=2.91, [NII]λ6583/Hα=0.17,and[SII]λλ6717,6731/Hα=0.09 place the object clearly into the domain of HII regions in Figs.1,2of Veilleux&Osterbrock(1987;VO87). This is also confirmed by the weakness(non-detection)of [OI]λ6300in the spectrum.Because theflux calibration using our standard stars is uncertain,we transformed it to the Jacoby(1989)m OIII calibration of the imaging photometry from Suprime-Cam (Arnaboldi et al.2002).This gives a total m OIII=25.7 magnitude for this object.Because of the[OIII]fil-ter width used in the emission line survey,thisflux in-cludes both the[OIII]λλ4959,5007lines,as well as the flux from the ing the spectrum to mea-sure the fraction of light in the continuum,we can correct to the magnitude in theλ5007line only:m5007=26.15. From the standard star–calibrated spectrum,we would get m5007=−2.5log F5007−13.74=25.1.The difference gives us a factor2.63,by which to rescale F5007to match the photometric calibration.The rescaled totalflux in the [OIII]λ5007˚A line is F5007=11.0×10−17erg s−1cm−2. With E(B-V)=0.4the dereddenedflux becomes F5007= 4.0×10−16erg s−1cm−2.Thefluxes in the other emis-sion lines can then be inferred from the dereddened line ratios in Table1;in particular the Hαflux becomes 3.9×10−16erg s−1cm−2.The measured V-band continuumflux from the spec-trum,when applying the same calibration and correcting for reddening,becomes F V=7.0×10−16erg s−1cm−2. The corresponding dereddened apparent magnitude is m V=24.2.Absolute magnitudes andfluxes will be com-puted for a nominal Virgo distance modulus of31.16,cor-responding to17Mpc distance(Tonry et al.2001).How-ever,we will sometimes keep the distance dependence by writing D=d17Mpc.The continuum m V=24.2then gives M V=−7.0.3.PHYSICAL PARAMETERSMetallicity:The high values of[OIII]/Hα=1.0and [OIII]/Hβ=2.9indicate subsolar metallicity.The VO87 diagrams are not ideal for estimating metallicities,as dis-cussed by Dopita et al.(2000),but from Figs.2,3of that paper we estimate Z≃0.4and an ionization parame-ter q≃4×107(log U=−2.9).Both very low and high metallicities are not consistent with their model re-sults and these line ratios.A more accurate determi-nation is in principle possible using the S23parameter, defined as S23=([SII]λλ6717,6731+[SIII]λλ9069,9532)/ Hβ=0.75,where we have used the theoretical ratio [SIII]λ9532/[SIII]λ9069=2.48.From the empirical cal-ibration of D´iaz&P´e rez-Montero(2000)we thenfind 12+log(O/H)≃8.08±0.2(about0.15to0.25solar with or without the depletion factor used by Dopita et al.2000). In our case,S23has additional uncertainties due to the weak[SII]line and the bright NIR sky lines,which might affect the[SIII]flux.In the following,we therefore adopt Z≃0.4.Temperature,density,ionization parameter:Un-fortunately,the S/N of the spectrum is not large enough to detect the weak[OIII]λ4363and[NII]λ5755lines used to determine electron temperature,nor do we have lines to determine the electron density.In the following we use T e=104K when needed.Estimates of the ionization parameter from the Sulfur lines,using equations(8)and(6)of D´iaz et al.(2000) give similar values to that from the VO87diagrams: [SII]λλ6717,6731/[SIII]λλ9069,9532≃0.53gives log U=−2.5,and[SII]λλ6717,6731/Hβ≃0.26and metallicity0.4 solar gives log U=−2.9.Luminosities:From above,the total V-band luminos-ity isL V=4πD2F V=2.4×1037d217erg s−1.(1) The Hαflux isL Hα=4πD2F Hα=1.3×1037d217erg s−1.(2) The total number of H-ionizing photons(Osterbrock1989) with recombination coefficientsαB andαeffHαfor T=104K isQ(H0)=2.96L Hα/hνHα=1.3×1049d217s−1.(3) Stellar mass and age of starburst:The ratio Q(H0)/L V decreases rapidly with the mass of the most massive surviving O stars,i.e.,the age of the starburst. Using the Starburst99model of Leitherer et al.(1999)for metallicity0.4solar and normal Salpeter IMF,we deter-mine an age of3.3Myr.Once this is known,the Lyman continuum luminosity can be used to infer the total mass in the corresponding starburst;we obtain400M⊙.The number of O stars involved is1–2,so these numbers must be considered as averages.Mass and size of gas cloud:We can estimate the total mass of ionized hydrogen from(Osterbrock1989) M HII=Q(H0)m p/[n eαB]=420n−1100d217M⊙,(4) where n100=n e/100cm−3and m p denote the electron density and proton mass.Around early O stars,the H Str¨o mgren radius isr HII= M HII(1+y+)4Gerhard et al. where y+≃0.1is the fraction by number of ionized he-lium.This is indeed much smaller than the photometricspatial resolution(about0′′.7=57d17pc).The columndensity isN HII=M HII/(πr2HII)=1.3×1021n1/3100d2/317cm−2.(6)From the inferred extinction and metallicity we may es-timate the intervening hydrogen column density from the local interstellar medium relation(Bohlin,Savage&Drake 1978),usingN(H)≃5.9×1021cm−2mag−1E B−V Z⊙/Z∼6×1021cm−2.(7) If the neutral hydrogen is in a much denser shell than the HII,then we can use the HII radius also to estimate its total massM H=πr2HII×2N(H)m p=3600M⊙.(8) The star formation efficiency would then be around10%.4.DISCUSSIONThe compact HII-region is located about3.′4(17kpc projected distance)north and0.′9(4.4kpc)west of NGC 4388,almost perpendicular to the disk plane of this galaxy and45◦away from the nearest part of the very extended emission-line region(VEELR)discussed by Yoshida et al. (2002).The radial velocity inferred from the emission lines is2670km s−1,whereas the galaxy has v r=2520km s−1. The near-coincidence of these numbers may indicate that the HII region is,or once was,part of the NGC4388sys-tem.From the large radial velocity relative to the Virgo center(v r∼1.8σVirgo)and from the Tully-Fisher distance of NGC4388(Yasuda et al.1997),both are probably falling through the cluster core.This HII region is powered by a small stellar associa-tion or star cluster,with an estimated mass of∼400M⊙and age of∼3Myr for a normal IMF.If it is a young cluster,it must have a radius smaller than that of the HII region(∼3.5pc).Clusters with these parameters have short relaxation times and dissolve by internal dynamical processes;within a few108yr the stars would be added to the diffuse stellar population nearby.The formation of such objects is thus possible far out-side the main star forming regions in galaxies.Perhaps the most plausible explanation for the observed position and velocity of the HII region relative to NGC4388is that it is already unbound and moving on a different orbit in the cluster potential.In this case we would be seeing intraclus-ter star formation.If on the other hand its true distance from NGC4388is comparable to the projected distance so that it is still bound,we would have discovered a small star-forming knot in the far outer halo of NGC4388far from any other star formation activity.This would have a velocity in the frame of the galaxy comparable to the cir-cular velocity(≃200km s−1)of NGC4388.In fact,in the Subarufield that contained our ICHII region,Arnaboldi et al.(2002)found a small sample of similar candidate objects,among which7candidates are located in the out-skirts of M86,at a distance of10–15kpc in a disturbed region which probably also contains diffuse Hαemission, and1such object in the outer parts of M84.What has triggered the recent onset of star formation in this remote cloud?Notice that with its radial velocity,it will have moved∼4kpc relative to the Virgo cluster,and ∼500pc relative to NGC4388,in the lifetime of the mas-sive stars.Thus these stars will essentially have formed in situ.A possible model could be that the cloud was com-pressed after entering the hot intracluster medium(ICM). While the typical pressure of the ICM(n e T∼104K/cm3) is far smaller than the internal pressure inferred from typ-ical HII region parameters(n e∼100/cm3;T∼104K),it could be comparable to that of the surrounding neutral or molecular cloud traced by the absorption.The ionized re-gion may be reexpanding into the surrounding cloud and into the ICM–the mass of the cluster is not large enough to bind the HII region.Reanalysing the Subarufield photometry,we have also detected a number of extended emitters in both[OIII]and Hα,with colors similar to the confirmed compact HII re-gion.These are peaks in more extended emissions,and lie at distances11–33kpc along the direction of the VEELR identified by Yoshida et al.(2002)towards the NE of NGC4388.These objects have relatively low excitation ([OIII]/Hα≃0.6,in agreement with Fig.6of Yoshida et al.2002),and show continuum emission near the limiting magnitude of the combined(V+R)image(Arnaboldi et al. 2002).Their half-light radii are similar in the combined continuum and in the[OIII]and Hαimages,which fur-ther supports the notion that these distant sources have underlying continuum emission,and are thus ionized by OB stars rather than by the nucleus of NGC4388.From the discussion of Yoshida et al.(2002)it is likely that this star formation is induced by the jet or ionization cone. Yoshida et al.(2002)proposed that the VEELR ob-served to the NE of NGC4388could be the debris of a past interaction.Although our ICHII region is2.6arcmin away from the nearest part of the VEELR,it could also be associated with such tidal debris.The gas in this ICHII region seems unlikely to come from ram pressure stripping of the gas in NGC4388,because the peculiar velocity of the ICHII region relative to the systemic velocity of the Virgo cluster is even larger than for NGC4388itself.(We thank B.Moore for this comment.)The total intracluster star formation rate(ISFR)es-timated from the candidates in thisfield is small,how-ever.Counting the8ICHII candidates near M86and M84plus3intracluster candidates,including that stud-ied here,and the6best extended candidates,gives a to-tal of17star-forming HII regions in the Subarufield.If the density of these objects is typical for the Virgo clus-ter core,these would correspond to∼103such objects throughout Virgo.If we adopt similar parameters to those inferred for the HII region here(conservatively),we ob-tain from these candidates a total ISFR of∼10times ∼400M⊙per3Myr,in a surveyed area of918arcmin2, i.e.,∼10−6M⊙arcmin−2yr−1.For comparison,the intra-cluster luminosity inferred from planetary nebulae in the Subarufield is∼107L B,⊙arcmin−2.However,it appears not impossible that at higher redshifts,when the environ-ment of infalling galaxies was more gas-rich,formation of stars directly from this intracluster gas could have been an important part of the origin of intracluster stars in Virgo. The existence of compact HII regions in Virgo,beyond its intrinsic interest,is relevant for a few other issues as well.Firstly,the massive stars ionizing the gas explode asIsolated HII Region in Virgo Cluster5type II supernovae and enrich the Virgo ICM with met-als.This process adds to the main metal content of the ICM,which is believed to have occured at high redshift when most of the stars in the cluster ellipticals and bulges formed(Renzini1999).From the ISFR inferred above,the present expected supernova rate is∼10−8arcmin−2yr−1. SN II from isolated star formation could provide the en-richment inferred for the Lyαclouds in Virgo(Tripp et al.2002).Because our ICHII region contains only1-2O stars,observations of the enrichment from such objects in isolated regions might give constraints on the element yields of individual supernovae.Newly formed stars from this material could then have a range of abundance ratios similar to old Galactic halo stars(Argast et al.2000). Secondly,compact HII regions may affect distance deter-minations with the planetary nebula luminosity function (PNLF,Jacoby et al.1992).The[OIII]luminosity of this ICHII region places it at the bright end of the luminosity function inferred from[OIII]emission line surveys.In a sufficiently deep offband control image its continuum light would be visible,leading to removal from the PN sample. However,this does require a deep image,and moreover there are a few HII candidates in the Subarufield for which the continuum is not detected.A few unrecognized such objects per galaxy or intraclusterfield could cause the dis-tance to thesefields to be underestimated.It is unclear whether this may account for the lower average distance inferred by the PNLF compared to surface brightnessfluc-tuations(Ciardullo et al.2002).Thirdly,if compact ICHII regions exist in galaxies gen-erally,they could be the birth places of distant B stars in the Galactic halo,some of which are too far from the disk to have been ejected from there into the halo(Con-lon et al.1992).Most likely,their birth places would be distant high velocity clouds.At this time,however,there is no evidence for star formation in galactic high-velocity clouds.We thank R.Scarpa for efficient help at UT4,J.Alcal´a for independently checking linefluxes,and A.Capetti for a helpful discussion.OG thanks the Swiss Nationalfonds for support under grant20-64856.01.This research has made use of the NASA extragalactic database.REFERENCESArgast,D.,Samland,M.,Gerhard,O.E.,&Thielemann,F.-K. 2000,A&A,356,873Arnaboldi,M.,et al.1996,ApJ,472,145Arnaboldi,M.,et al.2002,AJ,in pressBernstein,G.M.,et al.1995,AJ,110,1507Bicknell,G.V.,et al.2000,ApJ,540,678Bohlin,R.C.,Savage,B.D.,&Drake,J.F.1978,ApJ,224,132 Cardelli,J.A.,Clayton,G.C.,&Mathis,J.S.1989,ApJ,345,245 Ciardullo,R.,et al.2002,ApJ,577,31Conlon,E.S.,Dufton,P.L.,Keenan,F.P.,McCausland,R.J.H.,& Holmgren,D.1992,ApJ,400,273D´iaz,A.I.,P´e rez-Montero,E.2000,MNRAS,312,130D´iaz,A.I.,Castellanos,M.,Terlevich,E.,&Garc´ia-Vargas,M.L. 2000,MNRAS,318,462Dopita,M.A.,Kewley,L.J.,Heisler,C.A.,&Sutherland,R.S.2000, ApJ,542,224Feldmeier,J.J.2002,IAU Symp.209,Intracluster Planetary Nebulae,ASP,in press,astro-ph/0201452Ferguson,H.,Tanvir,N.R.,&von Hippel,T.1998,Nature,391,461 Gavazzi,G.,et al.2001,ApJ,563,L23Jacoby,G.H.1989,ApJ,339,39Jacoby,G.H.,et al.1992,PASP,104,599Leitherer,C.,et al.1999,ApJS,123,3Moore,B.,Lake,G.,Quinn,T.,&Stadel,J.1999,MNRAS,304, 465Okamura,S.,et al.2002,PASJ,in pressOsterbrock,D.E.1989,Astrophysics of Gaseous Nebulae and Active Galactic Nuclei,University Science Books,Mill ValleyQuilis,V.,Moore,B.,&Bower,R.2000,Science,288,1617 Renzini,A.1999,in Chemical Evolution from Zero to High Redshift,eds.J.Walsh,M.Rosa(Berlin,Springer),185Tonry,J.L.et al.2001,ApJ,546,681Tripp,T.M.,et al.2002,ApJ,575,697Veilleux,S.,&Osterbrock,D.E.1987,ApJS,63,295(VO87) Yasuda,N.,Fukugita,M.,Okamura,S.1997,ApJS,108,417 Yoshida,M.,et al.2002,ApJ,567,118。